Zinc Nitrate — Structure, Properties, and Industrial Uses
Fundamental Identity and Structural Nature of Zinc Nitrate
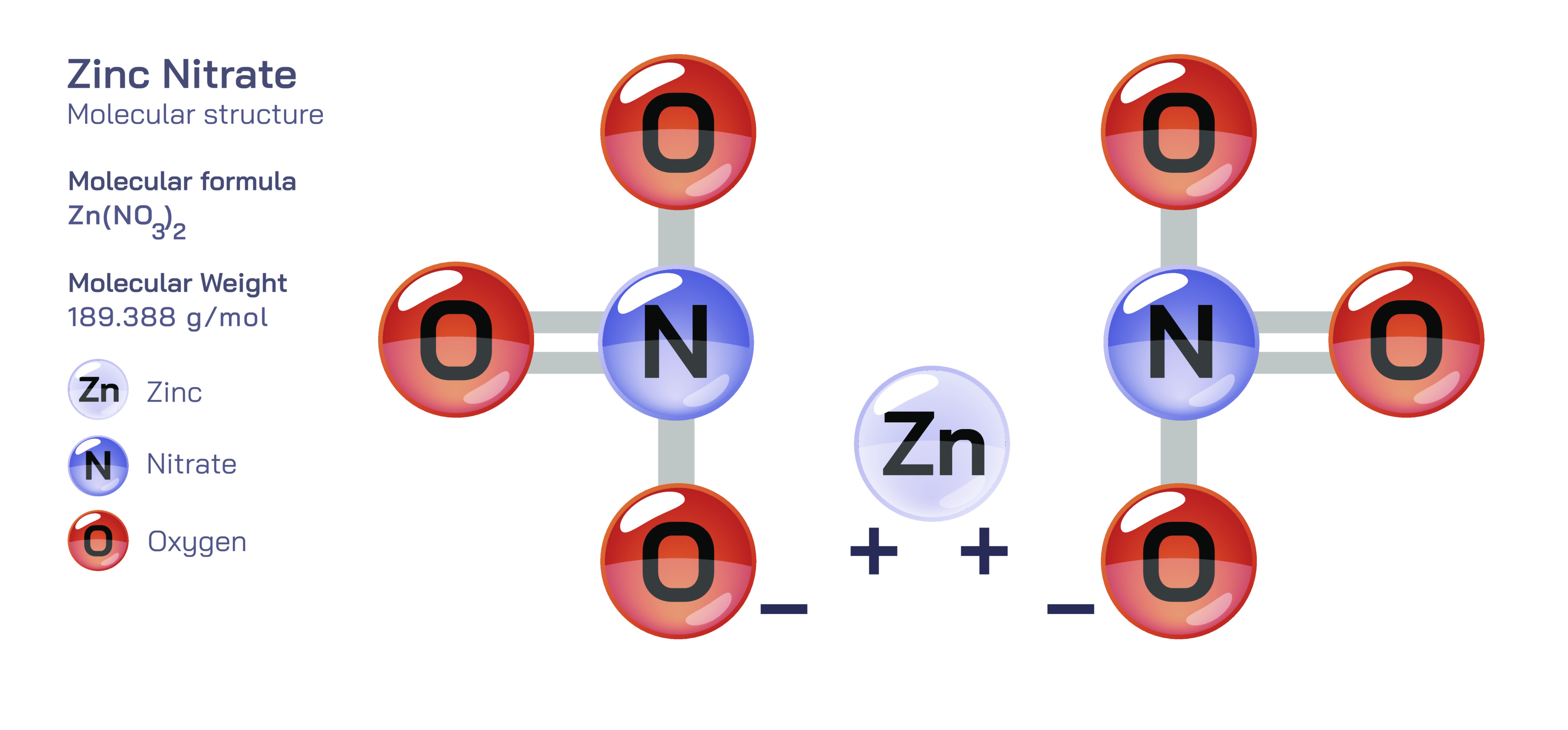
Zinc nitrate, commonly written as Zn(NO₃)₂, is a highly soluble inorganic salt composed of zinc cations and nitrate anions. In its pure form, it typically appears as a crystalline solid, often found in hydrated forms such as zinc nitrate hexahydrate (Zn(NO₃)₂·6H₂O). The presence of water molecules within the crystalline network plays a significant role in stabilizing the structure and influencing its physical and chemical properties. Structurally, the Zn²⁺ ion is coordinated with both nitrate ions and water molecules, forming complex arrangements that vary depending on hydration. The nitrate group, NO₃⁻, features a trigonal planar geometry with delocalized electron density across the three oxygen atoms, giving it notable resonance stability. These nitrate ions interact electrostatically with the zinc center, and in hydrated crystals, water molecules further coordinate with the metal ion, generating various octahedral coordination environments. This structural flexibility allows zinc nitrate to dissolve rapidly in water, disassociate into ions, and participate readily in chemical reactions, making it a versatile compound across laboratory, industrial, and materials science applications.
Chemical Properties and Reactivity Patterns of Zinc Nitrate
Zinc nitrate is defined by its strong ionic character and its readiness to ionize completely in aqueous solutions, forming Zn²⁺ and NO₃⁻ ions. The zinc ion, with its moderate Lewis acidity, readily forms complex ions with water, ammonia, and other ligands, enabling zinc nitrate to function as a useful precursor in coordination chemistry. The nitrate ion, while generally stable and unreactive in mild conditions, can participate in redox reactions under strong heating or in the presence of reducing agents. When zinc nitrate is heated, it undergoes decomposition, releasing nitrogen oxides and forming zinc oxide as a stable residue. This thermal decomposition is associated with the breakdown of the nitrate component, which yields gases such as nitrogen dioxide and oxygen. In solution, zinc nitrate behaves as a typical zinc salt, reacting with bases to form zinc hydroxide precipitates, which can then convert into zinc oxide upon heating. Because the nitrate ion does not form complex salts like sulfates or chlorides, solutions of zinc nitrate maintain clarity and high mobility, making it ideal for chemical syntheses, electrochemical applications, and processes that require transparent ionic solutions. The compound’s oxidizing potential is mild compared to other nitrates, but its ability to release nitrogen oxides under heat requires careful handling, particularly in industrial-scale operations.
Physical Characteristics and Solubility Behavior
Physically, zinc nitrate is best known for its high level of solubility in water and other polar solvents. The hydrated crystals of zinc nitrate hexahydrate are typically colorless, deliquescent, and capable of absorbing moisture from the air. This makes storage conditions important, as prolonged exposure to humidity can lead to gradual dissolution of the solid material. The crystals melt at relatively low temperatures due to the release of coordinated water molecules, after which the compound decomposes rather than forming a stable anhydrous melt. The appearance of zinc nitrate is often described as glassy or transparent, reflecting the uniformity of its crystalline lattice in hydrated forms. Its high solubility is a key reason for its usefulness in preparing uniform zinc-containing coatings, catalyst precursors, and solutions for analytical and electrochemical processes. The ionic mobility of Zn²⁺ and NO₃⁻ in solution also contributes to its role in metal plating and conductive applications. Overall, the compound’s physical traits—particularly its solubility and deliquescent nature—make it easy to handle in solution but challenging to preserve in solid form without controlled environmental conditions.
Industrial and Laboratory Uses Across Chemical Sectors
Zinc nitrate holds significant value in a range of industries, most notably in chemical manufacturing, materials science, agriculture, and catalysis. One of its primary uses is as a precursor in the production of zinc oxide through controlled thermal decomposition. The resulting zinc oxide is used in rubber vulcanization, ceramics, pigments, pharmaceuticals, and UV-absorbing materials. In the coatings industry, zinc nitrate solutions are used for zinc-based surface treatments, corrosion inhibitors, and metal finishing processes that require even distribution of zinc ions. The nitrate form enhances solution clarity, enabling uniform deposition in electroplating, battery component manufacturing, and thin-film preparation. In agriculture, zinc nitrate solutions act as micronutrient fertilizers that correct zinc deficiency in soils and crops. Due to its solubility, zinc from this compound is readily absorbed by plant roots or foliage in foliar sprays, supporting enzyme function, growth regulation, and overall plant health.
In catalysis, zinc nitrate serves as a precursor for producing zinc-based catalysts used in organic transformations, redox processes, and environmental remediation. It is also utilized in laboratory synthesis to prepare complex zinc salts, metal–organic frameworks, and nanoparticle materials. In pyrotechnics, zinc nitrate appears as an oxidizer in certain formulations, contributing to combustion efficiency and coloration effects. In analytical chemistry, zinc nitrate is used in testing protein content and as a reagent in qualitative inorganic analysis. Its versatility across so many chemical sectors arises from the reliability of its ionic behavior, its ease of dissolution, and its ability to form predictable, stable end-products when decomposed or reacted under controlled conditions.
Environmental and Safety Considerations
Like all nitrates, zinc nitrate must be handled with respect for its environmental and biological impact. Although zinc is an essential micronutrient for plants, animals, and humans, excessive zinc concentrations can become toxic to aquatic organisms and soil ecosystems. Careful dosing in agricultural use prevents nutrient imbalance or runoff into waterways. The nitrate component poses additional environmental concerns, as heavy nitrate release contributes to eutrophication—excess nutrient loading that disrupts aquatic systems. Industrial waste streams containing zinc nitrate therefore require proper treatment to remove or neutralize both zinc and nitrate ions before disposal. When heated, zinc nitrate produces nitrogen oxides, which are air pollutants and require controlled emission handling. Although the compound itself is not highly hazardous under normal handling, its oxidizing nature and decomposition products necessitate protective measures in industrial environments. With responsible management, zinc nitrate remains a safe and effective chemical for a wide range of beneficial applications.
Illustrating the Structure and Chemical Behavior of Zinc Nitrate
Scientific illustrations of zinc nitrate often highlight the trigonal planar geometry of the nitrate ion, showing how the three oxygen atoms symmetrically surround the central nitrogen atom. Diagrams also depict zinc ions surrounded by water molecules or oxygen atoms from nitrate groups, forming octahedral coordination complexes in hydrated crystals. Structural visuals may show the orderly arrangement of molecules in the hexahydrate form, emphasizing how water molecules stabilize the overall lattice. Industrial illustrations commonly depict zinc nitrate solutions being used in metal plating baths, agricultural sprayers, or catalyst preparation reactors, visually linking its chemical structure to its real-world functions. Thermal decomposition diagrams often show the transformation from zinc nitrate to zinc oxide, along with the release of nitrogen oxides. Through these representations, the connection between the compound’s molecular geometry, properties, and diverse applications becomes clear, demonstrating why zinc nitrate is recognized as one of the most adaptable and useful zinc salts in chemistry.