Zinc Sulfate — Structure, Uses, and Chemical Properties
Fundamental Identity and Structural Composition of Zinc Sulfate
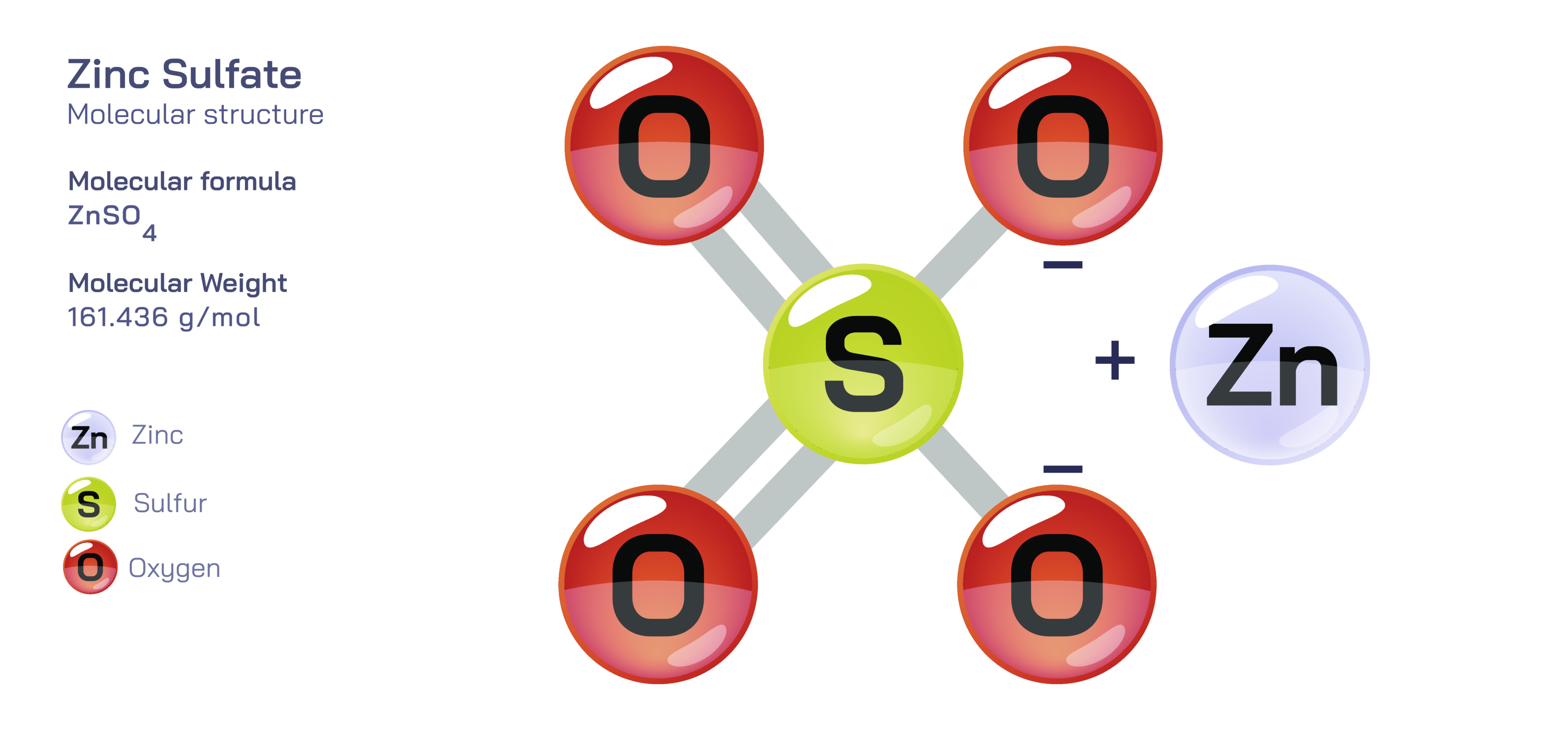
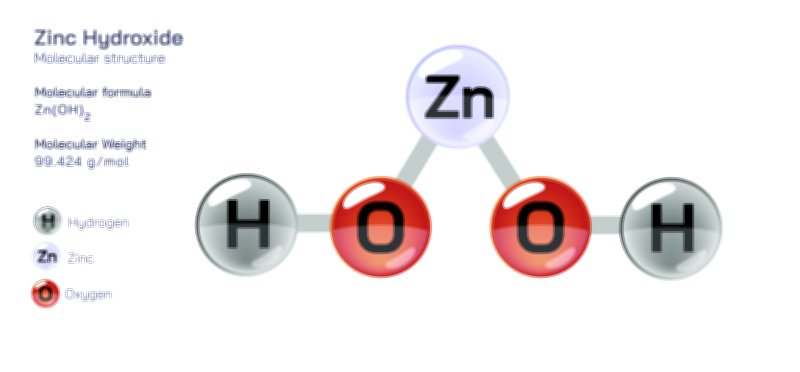
Zinc sulfate, commonly expressed by the formula ZnSO₄, is an important inorganic compound that occupies a central place in chemical manufacturing, nutrition, agriculture, medicine, and numerous industrial processes. While its anhydrous form exists as a simple salt consisting of zinc cations and sulfate anions, it more commonly appears in various hydrated forms, the most prevalent being zinc sulfate heptahydrate (ZnSO₄·7H₂O). This hydrated version forms transparent, crystalline solids that dissolve readily in water, reflecting the strong ionic nature of the compound. Structurally, the zinc ion exists as Zn²⁺, tightly coordinated with oxygen atoms either from water molecules or from the sulfate ion. The sulfate group, SO₄²⁻, possesses a tetrahedral geometry, with a central sulfur atom surrounded by four oxygen atoms. In the heptahydrate, zinc typically coordinates with a combination of sulfate oxygens and water molecules, forming an octahedral complex that stabilizes the crystalline lattice. This balance between ionic bonding, water coordination, and sulfate geometry gives zinc sulfate its characteristic solubility, reactivity, and functional versatility. Its structure is robust yet adaptable, enabling it to play critical roles across industrial, pharmaceutical, and biological systems.
Chemical Properties and Behavior in Various Environments
Chemically, zinc sulfate exhibits a strong ionic nature, dissociating readily into its constituent ions when dissolved in water. This dissociation enables zinc to interact with biological systems, soil chemistry, and industrial reagents, making the compound both reactive and useful. The Zn²⁺ ion is moderately acidic in aqueous solutions, often participating in hydrolysis reactions that form zinc hydroxide or other zinc-based complexes depending on pH. In acidic solutions, zinc sulfate remains stable and highly soluble, supporting applications where zinc must be delivered in bioavailable or reactive form. In basic environments, however, the zinc ion can precipitate as zinc hydroxide or form additional complex ions, illustrating how pH strongly influences the compound's behavior. Heating zinc sulfate leads to dehydration, with stepwise release of water molecules that ultimately produce the anhydrous form. Further heating causes decomposition into zinc oxide and sulfur trioxide, a transformation leveraged in industrial zinc chemistry. Zinc sulfate does not act as an oxidizing agent despite containing sulfur in a high oxidation state; instead, it remains largely inert except in conditions where its ionic components undergo precipitation or thermal decomposition. These chemical behaviors underscore zinc sulfate’s reliability and controlled reactivity, characteristics that have made it a staple compound across many fields.
Physical Characteristics and Their Functional Significance
Zinc sulfate typically appears as a colorless, crystalline solid with a slightly astringent taste. The heptahydrate form crystallizes in long, prismatic structures that dissolve readily in water, forming clear, strongly ionic solutions. Its solubility makes it particularly valuable for applications requiring rapid dispersion and bioavailability. The compound exhibits moderate density and stability under dry conditions, though it can lose water molecules upon exposure to heat or low humidity. The hydrated forms melt at relatively low temperatures, where crystal water is released in stages. This water content plays an important role in industrial handling, as hydrated zinc sulfate flows well, dissolves easily, and is simpler to store and ship compared to the anhydrous form. The optical clarity of its crystals also makes zinc sulfate useful in laboratory crystallography and chemical demonstration settings. These physical traits—solubility, structural stability, and hydration behavior—support its widespread functionality in chemical formulations, nutrient supplements, industrial processes, and environmental remediation systems.
Biological, Agricultural, and Industrial Uses of Zinc Sulfate
Zinc sulfate is extensively used across biological, agricultural, and industrial sectors due to its ability to supply bioavailable zinc and its interactions with various chemical processes. In agriculture, zinc sulfate serves as one of the most important micronutrient fertilizers, correcting zinc deficiencies in crops such as wheat, rice, maize, and citrus. Zinc is essential for enzymatic function, protein synthesis, and root development, and its deficiency leads to stunted growth, leaf discoloration, and reduced yields. By providing zinc in a soluble, easily absorbed form, zinc sulfate restores soil nutrient balance and supports healthy crop development. In medicine and nutrition, zinc sulfate is used as a dietary supplement to treat zinc deficiency in humans, supporting immune function, wound healing, and cell growth. It is incorporated into oral solutions, tablets, and eye drops, and it also plays a role in topical astringent preparations. Industrially, zinc sulfate is used in electroplating, textile processing, leather treatment, and as a component in the production of lithopone pigments. It participates in coagulation processes during water purification, where it helps precipitate impurities and improve clarity. In laboratory chemistry, zinc sulfate serves as a reagent for testing the presence of proteins, as it causes protein precipitation, and it is used in flotation processes for mineral separation. Across all these fields, zinc sulfate’s reliability, solubility, and chemical accessibility make it an indispensable compound.
Environmental and Chemical Significance in Natural Systems
In environmental chemistry, zinc sulfate plays a role in soil conditioning, water treatment, and ecological nutrient balancing. Zinc is a trace element essential for organisms, and zinc sulfate provides a way to restore depleted soils without introducing harmful contaminants. In wastewater treatment, zinc sulfate can assist in removing unwanted heavy metals by participating in precipitation reactions or by adjusting ionic balances in solution. Its controlled solubility allows it to be used in environmental remediation efforts where mobilization or immobilization of ions must be carefully managed. In natural systems, zinc sulfate does not persist in a harmful state under normal conditions, as zinc eventually integrates into soil minerals or biochemical cycles while sulfate disperses harmlessly through natural water pathways. However, excessive concentrations can lead to toxicity, underscoring the need for responsible application in agriculture and industry. Understanding zinc sulfate’s environmental behavior ensures that its beneficial effects are maximized while potential ecological impacts remain controlled and minimal.
Illustrating the Structure and Functional Behavior of Zinc Sulfate
Scientific illustrations of zinc sulfate often highlight the sulfate group’s tetrahedral geometry, showing the sulfur atom at the center bonded to four oxygen atoms arranged symmetrically. Diagrams also depict the zinc ion coordinating with water molecules or sulfate oxygen atoms, representing the octahedral complex typical of the heptahydrate form. Visual representations may show the crystalline structure of the hydrated salt and depict how water molecules are integrated into the lattice. Industrial illustrations frequently highlight its use in fertilizers, water treatment tanks, electroplating baths, or pharmaceutical formulations, providing a clear link between its molecular behavior and real-world functions. These diagrams help explain how zinc sulfate dissolves, how its ions behave in soil or biological tissues, and how heating causes dehydration and decomposition into other useful zinc compounds. Through such illustrations, the connection between structure, chemical properties, and widespread applications becomes visually intuitive, emphasizing the compound’s importance across science, health, and industry.