Moseley’s Law and the Atomic Number Relationship in Physics
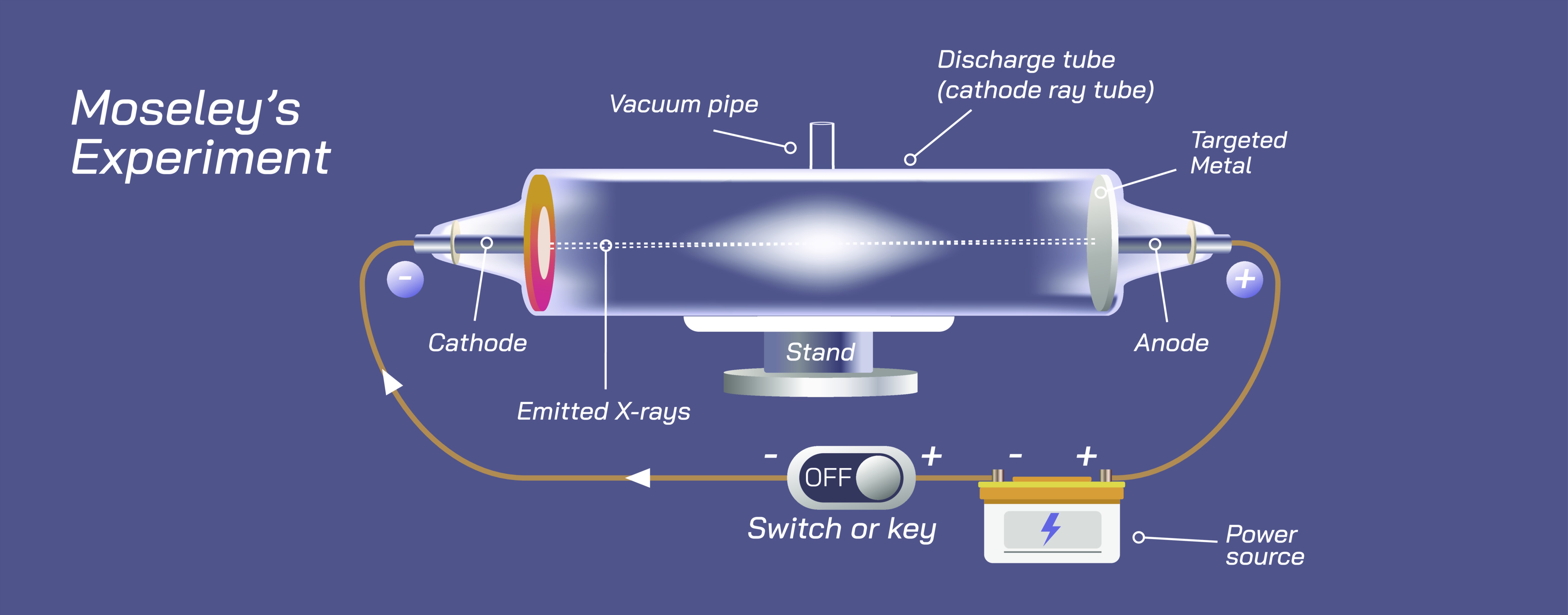
Moseley’s law stands as one of the most transformative discoveries in modern physics because it redefined our understanding of the periodic table not as a collection of elements arranged by mass or chemical similarity, but as a system governed by a deeper physical quantity—the atomic number. Before the early twentieth century, scientists believed that the identity of an element was determined primarily by its atomic weight, and the periodic table was organized accordingly. This organization was almost correct but riddled with puzzling inconsistencies: some elements appeared to be in the wrong order by mass yet clearly belonged in certain chemical families, and the existence of unexplained gaps suggested missing elements but offered no systematic way to predict them. The breakthrough came in 1913 when physicist Henry Moseley used X-ray spectroscopy to study the radiation emitted by atoms. By measuring the frequencies of characteristic X-rays emitted when electrons transitioned to the innermost shells of various elements, Moseley discovered a simple, elegant, and universal pattern: the square root of the emitted X-ray frequency increased linearly with the atomic number of the element. This relationship, later called Moseley’s law, revealed that the atomic number was not just a label but a physical quantity reflecting the positive charge of the nucleus. In a single discovery, Moseley provided the missing foundation that linked the structure of the atom to the structure of the periodic table.
The significance of this discovery becomes clear by retracing the structure of atomic energy levels. When atoms are bombarded with high-energy electrons, inner-shell electrons can be ejected from their orbitals. Electrons from higher shells quickly fall into the newly created vacancies, and in doing so they release discrete amounts of energy in the form of X-ray photons. The energy—and thus the frequency—of these emitted photons depends on the strength of the attraction between the electrons and the nucleus. Moseley found that this attraction is determined by the number of positive charges in the nucleus, meaning the number of protons. Because protons exert stronger pull on inner-shell electrons as their quantity increases, the characteristic X-ray frequency increases in a predictable pattern across elements. The connection between frequency and atomic number was not a coincidence tied to chemical behavior; it was the direct physical manifestation of the structure of the atom. For the first time in history, the periodic table could be grounded not in macroscopic chemical reactivity but in the microscopic structure of the atomic nucleus.
Moseley’s law did more than correct a misunderstanding—it revolutionized the periodic table. By ranking elements according to their atomic number rather than atomic weight, it allowed every chemically related group to fall naturally into place. The law also identified specific atomic numbers that had no known corresponding elements at the time, proving that the periodic table contained true gaps waiting to be filled. The predicted elements—later confirmed as technetium, promethium, rhenium, and others—were eventually discovered exactly where the law anticipated. Moseley’s work also showed that some questionable elements believed to exist did not correspond to any missing atomic numbers and therefore likely did not exist. The periodic table transformed from a best-fit model built on chemical observation into a fully predictive structure grounded in nuclear physics. It was no longer a catalog of empirical discoveries but a map of nature’s fundamental building blocks.
A deeper scientific meaning began to emerge from this realization. Atomic number, not atomic mass, defines the identity of an element because the atomic number determines the number of protons in the nucleus, and the number of protons dictates the arrangement and behavior of electrons. It is the electron configuration that determines an element’s chemical properties, so the chemical behavior seen across the periodic table is ultimately a ripple effect of nuclear charge. Moseley therefore provided the link in the chain that connects chemistry to physics. The periodic repetitions of valence electron structures, which define trends in reactivity and bonding, originate from the discrete increases in nuclear charge that drive the structure of electron shells. In a broader sense, this discovery demonstrated that chemistry is a direct consequence of atomic physics, not an independent field governed by unexplained patterns.
The experimental clarity of Moseley’s law helped settle longstanding scientific debates that had resisted resolution through chemistry alone. One such debate involved the placement of argon and potassium. Based purely on atomic masses, potassium should have preceded argon, but their chemical behavior made that placement impossible. Moseley’s measurements revealed that argon has an atomic number of 18 and potassium has an atomic number of 19, confirming that order in the table must follow atomic number rather than atomic weight. The same approach correctly resolved the placement of cobalt and nickel, another pair whose masses seemed to follow the reverse of their chemical behavior. In each case, X-ray frequency—not mass—revealed the true identity of the element. Through this insight, Moseley supplied a principle that chemists had observed intuitively for decades without being able to explain: periodicity originates from nuclear charge.
The impact of the discovery reached beyond the laboratory and extended into global scientific development. Before Moseley’s law, atomic number was merely a counting system with no empirical foundation. After his discovery, atomic number became a measurable physical property, giving scientists a step-by-step numerical sequence extending across all known and yet-to-be-discovered elements. The table now appeared as a landscape of quantized nuclear structures, and the question of how many possible chemical elements could exist shifted from philosophical speculation to scientific exploration. The existence of transuranium elements—those beyond uranium—later became a natural consequence of extending nuclear charge beyond the heaviest naturally occurring atoms. The structure of the periodic table could now expand logically rather than heuristically.
Although Moseley’s law is rooted in experimental physics, its influence stretches deeply into quantum theory. The measured X-ray frequencies reflect the energy differences between electron orbitals, and those energy levels are determined by the solved equations of quantum mechanics for the hydrogen-like behavior of inner-shell electrons. The linear relation between the square root of X-ray frequency and atomic number emerges because the energy of an inner-electron transition depends on the effective nuclear charge seen by that electron. Quantum mechanics later confirmed the mathematical basis for Moseley’s findings: the frequency of emitted radiation corresponds to the square of the atomic number after accounting for electron shielding. Moseley identified this pattern experimentally before quantum mechanics explained it theoretically, making the discovery an early triumph of physical measurement guiding theoretical understanding.
The broader meaning of Moseley’s discovery becomes even clearer when the periodic table is viewed not simply as a chart of elements, but as a structural map of matter across the universe. Every atom in stars, planets, living organisms, and industrial materials adheres to the sequence of atomic numbers that Moseley helped define. The identity of iron in the Earth’s core, the oxygen we breathe, the silicon in computer chips, and the gold used in electronics is governed by the same physical principle revealed by X-ray spectra: the nucleus defines the atom. The number of protons controls the electric field within the atom, the electric field shapes the electron arrangement, and the electron arrangement determines chemical behavior. The recurring cycles of the periodic table, so long observed through empirical chemistry, find their causal origin in the nuclear physics that Moseley uncovered.
27. His death occurred at a time when the world was only beginning to recognize the significance of his discovery, and many historians agree that he might have achieved a Nobel Prize had he lived longer. Yet even without further work, the implications of Moseley’s law were powerful enough to reshape the entire foundation of atomic science. The connection between atomic number and nuclear charge remains one of the most important pillars of modern chemistry and physics, demonstrating that scientific progress often depends not only on theory but on precise observation of nature.
Moseley’s law remains a vivid reminder that the behavior of matter at the macroscopic scale—the diversity of elements, the structure of molecules, the reactions that sustain life and technology—is governed at the smallest scale by quantized properties of the atomic nucleus. The law did not merely fix the periodic table; it exposed the underlying reality that the universe is built from a systematic progression of nuclear charges, each generating distinct electron structures and chemical identities. By tying X-ray frequencies to atomic number, Moseley uncovered a relationship that bridges spectroscopy, nuclear structure, and chemical periodicity. It shows that order in the natural world is not arbitrary but structurally rooted in the physics of the atom. Through that insight, Moseley provided one of the clearest examples in scientific history of how observing a simple pattern in measurable data can unlock the organizing principle of the natural world.