Protein synthesis is one of the most fundamental biochemical processes sustaining life, and the elongation phase of translation is the driving engine behind polypeptide formation. During elongation, the ribosome reads the mRNA codons one by one and incorporates their encoded amino acids into a growing peptide chain. Although the process unfolds rapidly and continuously inside a cell, the underlying molecular coordination is precise, energy-dependent and tightly regulated to ensure accuracy and efficiency. The translation elongation cycle reflects the interplay of the ribosome, transfer RNA molecules, elongation factors and GTP hydrolysis, working together to convert genetic information into structured proteins.
Elongation begins once the first aminoacyl-tRNA has been positioned in the ribosome’s P site during initiation. The ribosome contains three functional tRNA-binding sites aligned across the mRNA: the A (aminoacyl) site, the P (peptidyl) site and the E (exit) site. Each cycle of elongation recruits a new aminoacyl-tRNA into the A site, catalyzes peptide bond formation and shifts the ribosome so the growing peptide chain moves from the A site to the P site. This process repeats until a termination codon brings synthesis to its conclusion.
The arrival of the next aminoacyl-tRNA marks the beginning of an elongation round. In eukaryotes, incoming aminoacyl-tRNAs are escorted by eEF-1α, a translation factor bound to GTP. Together they form a ternary complex that screens codons and delivers only the tRNA whose anticodon complements the mRNA sequence exposed in the A site. When the correct match is found, GTP hydrolysis occurs, altering the conformation of eEF-1α and releasing the aminoacyl-tRNA into the A site. The dissociated factor leaves as eEF-1α-GDP, which is not yet ready for another delivery cycle. To maintain high rates of translation, GDP must be exchanged for GTP, a reaction mediated by eEF-1βγ, a guanine nucleotide exchange factor that reactivates eEF-1α for additional delivery events. Without this recycling step, elongation would quickly slow and stall.
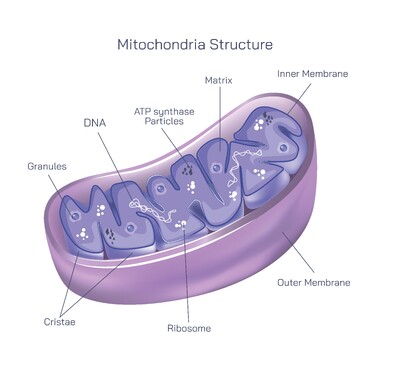
With an aminoacyl-tRNA properly positioned in the A site, the ribosome is prepared for peptide bond formation. The amino group of the incoming amino acid attacks the carbonyl carbon of the peptide attached to the tRNA located in the P site. This reaction is catalyzed not by proteins but by the ribosomal RNA itself. The 60S subunit contains the peptidyltransferase center, a functional region of the large 28S rRNA that behaves as a ribozyme. This ribozyme activity forms the backbone of polypeptide synthesis, demonstrating that RNA still carries catalytic capability rooted in ancient biology. Once the new peptide bond is created, the entire growing chain now resides on the tRNA positioned in the A site.
To continue elongation, the ribosome must move — this is the process of translocation. The tRNA holding the new polypeptide must shift from the A site to the P site, the previous P-site tRNA must move to the E site for release, and the ribosome must advance exactly three nucleotides along the mRNA to place the next codon into the A site. Translocation is not spontaneous; it is driven by eEF-2, a GTP-dependent molecular motor. eEF-2 binds to the ribosome, hydrolyzes GTP and induces a conformational shift that moves the ribosomal subunits relative to the mRNA. Once the ribosome reaches its new position, eEF-2 is released and the A site is ready for the next aminoacyl-tRNA. This step resets the elongation cycle.
2. When eEF-2 is phosphorylated, it loses activity and translocation slows. Phosphorylation is catalyzed by eEF-2 kinase (eEF-2K), a Ca²⁺/calmodulin-dependent protein kinase also known as CaMKIII. During periods of high muscular activity, intracellular Ca²⁺ levels rise and activate eEF-2K, slowing elongation so ATP consumption does not outpace cellular energy availability. eEF-2K itself is regulated by multiple pathways, including mTORC1, which represses eEF-2K to promote protein synthesis under nutrient-rich conditions, and AMPK, which activates eEF-2K under metabolic stress.
This sophisticated regulatory network illustrates that translation elongation is not a repetitive mechanical process, but a dynamic system reflective of a cell’s internal state. When nutrients are abundant and growth signals are active, elongation proceeds rapidly to accommodate protein turnover and cell expansion. When energy is scarce or stress signals are present, elongation is slowed or suspended to preserve ATP. The ribosome and elongation factors therefore act not only as molecular machines but also as metabolic sensors that respond to cellular physiology.
Despite its speed, elongation maintains remarkable fidelity. The ribosome monitors codon–anticodon pairing not only during tRNA delivery but also during selection for peptide bond formation. Incorrect tRNAs are rejected long before incorporation, minimizing translation errors. The cycle continues, adding amino acids sequentially with precision, until the ribosome encounters a stop codon and the termination machinery releases the completed protein.
The translation elongation cycle exemplifies molecular efficiency — rapid, repetitive, energetically powered and highly accurate. It converts information written in the linear language of mRNA into the three-dimensional functional architecture of proteins. The complexity of eEF-1α, eEF-1βγ, peptidyltransferase activity and eEF-2-mediated translocation reflects how evolution refined a single biochemical process into a regulated engine that drives growth, adaptation and survival in every living cell.