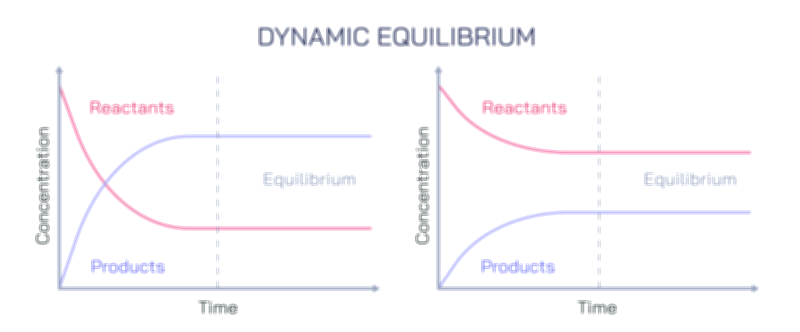
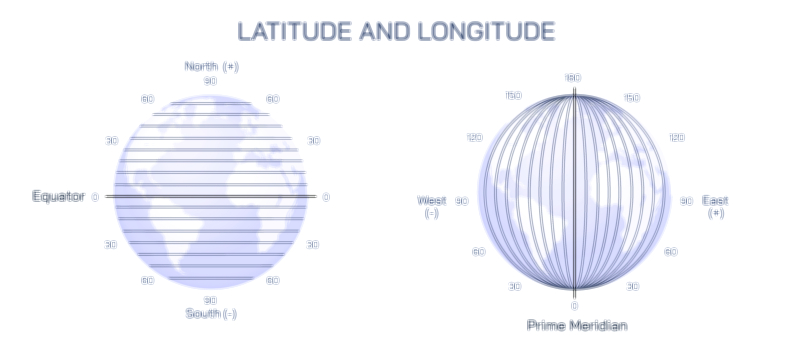
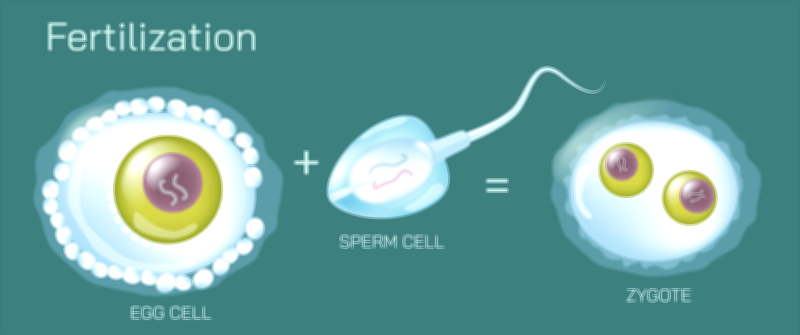
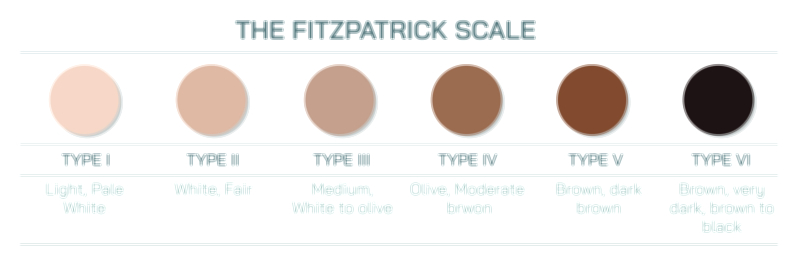
Litmus Paper and Color Change Test Vector Illustration Showing Acid and Base Identification Through Indicator Reaction
Litmus paper is a widely used chemical indicator that allows for the rapid identification of acidic or basic solutions. Derived from natural dyes, it changes color when exposed to solutions of different pH values, providing a simple and visual method for distinguishing between acids and bases. A vector illustration of the litmus paper test typically depicts red and blue litmus strips, test solutions, and resulting color changes, providing learners with a clear and educational understanding of the principle behind pH indicators. By integrating visual demonstration with chemical reasoning, such illustrations make the process of acid-base identification accessible, practical, and visually engaging.
At the core of the illustration is a set of litmus papers—one red and one blue—positioned alongside test solutions labeled for acids and bases. Red litmus paper is naturally red and is used to test for basic solutions, turning blue when exposed to a base. Conversely, blue litmus paper remains blue in neutral or basic solutions but turns red when exposed to an acidic solution. Vector diagrams typically include arrows indicating the placement of the paper into the solution, emphasizing the direction of the test and the interaction between the paper and the solution. Labels highlight the initial color of the paper, the type of solution, and the resulting color change, reinforcing the cause-and-effect relationship that underlies the indicator reaction.
The illustration often includes test tubes or beakers containing acidic and basic solutions, with droplets shown interacting with the litmus paper. For example, a vector diagram may depict a red litmus strip dipped into a beaker labeled “NaOH solution” and changing to blue, demonstrating its response to a base. Similarly, a blue litmus strip may be shown turning red when immersed in a solution labeled “HCl,” indicating an acid. Color coding, shading, or gradient overlays enhance the visibility of the transformation, making the result immediately comprehensible.
Vector diagrams may also illustrate neutral solutions, where litmus paper shows no color change. A beaker labeled “distilled water” or “neutral solution” can show red litmus remaining red and blue litmus remaining blue, visually reinforcing the concept of neutrality and providing a reference point for comparison. Arrows or dashed lines may indicate the expected reaction outcome in neutral solutions, emphasizing that color change occurs only under acidic or basic conditions.
Additional elements often depicted in the illustration include labels for the type of indicator, pH range, and chemical environment. Red and blue litmus papers may be marked with their corresponding pH sensitivity, showing that red litmus responds to pH above 7 (base) and blue litmus to pH below 7 (acid). Vector diagrams may include magnified sections of the litmus fibers to indicate where the dye interacts with hydrogen ions (H⁺) in acids or hydroxide ions (OH⁻) in bases, providing insight into the chemical mechanism behind the color change.
Vector illustrations frequently incorporate step-by-step procedural representation. For instance, the diagram may show sequential panels: first displaying the original color of the litmus paper, then dipping it into the solution, and finally showing the resulting color after the reaction. Arrows indicate the process direction, making it easy for learners to follow the testing procedure. Labels such as “Dip red litmus in base → turns blue” and “Dip blue litmus in acid → turns red” provide clear textual reinforcement, enhancing educational clarity.
The illustration may also depict safety and practical considerations. For example, gloves or a small dropper can be shown to indicate proper handling of test solutions, while arrows point to the litmus strip avoiding contamination from other substances. This visual guidance emphasizes laboratory best practices while maintaining focus on the indicator reaction.
By combining litmus paper, solution types, color change outcomes, and procedural steps, a vector illustration provides a comprehensive understanding of how acids and bases can be quickly identified using a simple chemical indicator. It visually demonstrates the responsiveness of red and blue litmus to chemical environments, clarifies the concept of pH sensitivity, and connects the theoretical principle to practical laboratory application.
Ultimately, a vector illustration of the litmus paper and color change test conveys the relationship between acids, bases, and chemical indicators, showing how hydrogen ion concentration directly affects the observable color change in litmus paper. Through labeled solutions, directional arrows, color-coded strips, and stepwise demonstration, the diagram transforms the abstract concept of pH measurement into an intuitive and educational visual tool, enabling learners to understand, perform, and interpret acid-base tests confidently.