ØÉÆØÉ©ØÉØØÉóØÉ«ØÉ” ØÉéØÉÜØɽØÉøØÉ©ØɦØÉÜØÉŁØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉłØÉ©ØɦØÉóØÉ£ ØÉüØÉ©ØɦØÉØØÉóØɦØÉĀ, ØÉÆØÉ©ØÉźØÉ«ØÉŁØÉóØÉ©Øɦ ØÉéØÉĪØÉ×ØÉ”ØÉóØɼØÉŁØɽØÉ▓ ØÉÜØɦØÉØ ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉŗØÉ©ØɦØÉĀ ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
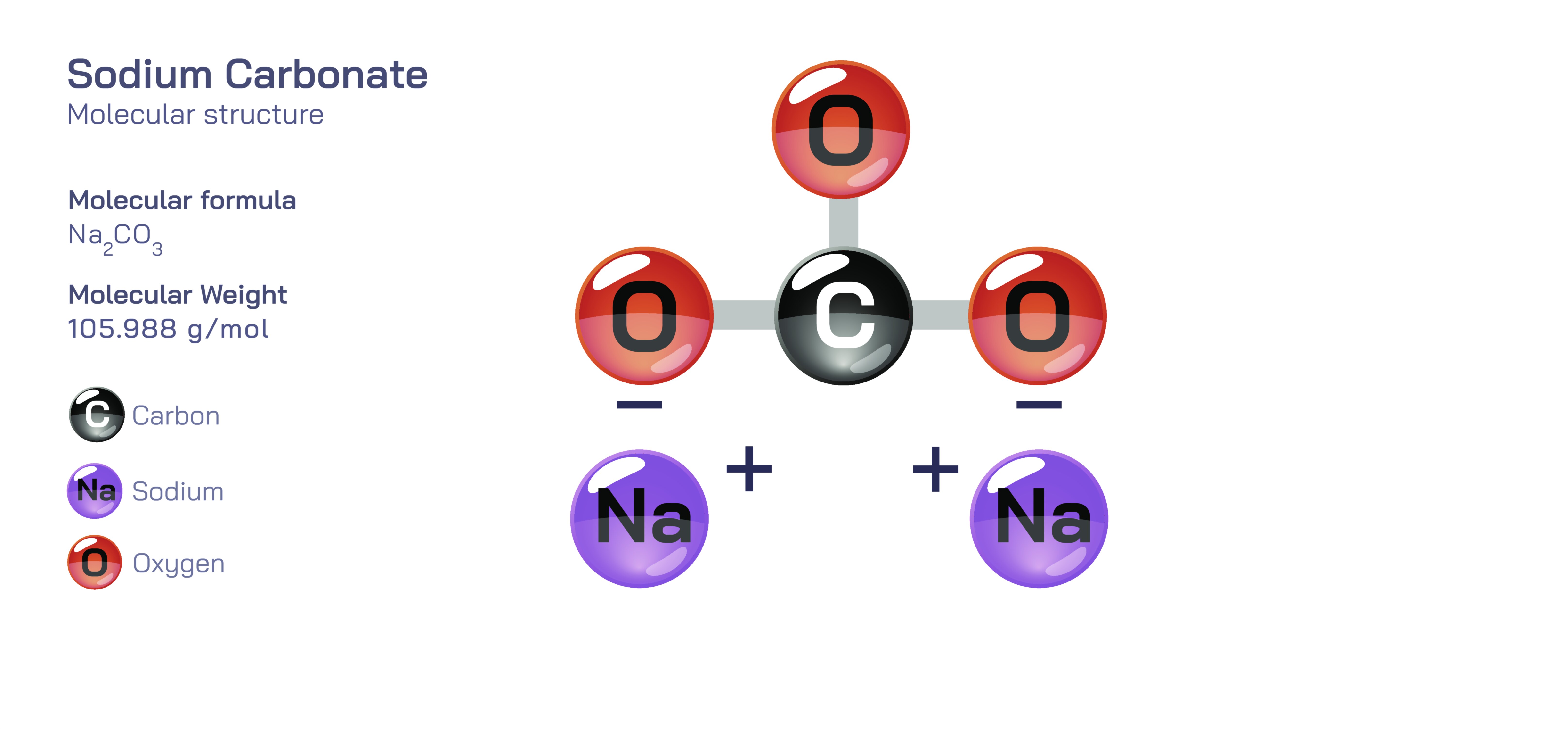
Sodium carbonate, often represented by the chemical formula NaŌééCOŌéā and commonly known as washing soda or soda ash, is one of the most important inorganic compounds in chemistry and industry. Although it appears as a simple white crystalline solid, its behaviour and structural identity are rooted in a remarkable interplay between ionic bonding, resonance, solubility, acidŌĆōbase equilibria and thermal stability. Understanding sodium carbonate requires exploring both the carbonate ion, which displays complex internal bonding, and the sodium cations, which provide ionic stability without interfering chemically. Every property associated with sodium carbonateŌĆöits strong alkalinity in water, its usefulness in glassmaking, its water-softening ability, and its role in acidŌĆōbase reactionsŌĆöcan be traced back to the molecular structure of the carbonate ion and its interaction with sodium ions and water. Because of these structural and functional relationships, sodium carbonate stands as an excellent model compound in chemistry education, exemplifying how atomic arrangement dictates macroscopic behaviour.
The structural core of sodium carbonate lies in the carbonate ion, COŌéā┬▓Ōü╗. This polyatomic ion contains one carbon atom centrally bonded to three oxygen atoms. Although typical Lewis diagrams often show one carbonŌĆöoxygen double bond and two carbonŌĆöoxygen single bonds, the actual structure of COŌéā┬▓Ōü╗ cannot be represented by any single arrangement. Instead, the negative charge and the extra pair of electrons are distributed evenly across the bonds through resonance, making all three CŌĆōO bonds equal in length and energy. This resonance stabilization gives the carbonate ion exceptional structural strength, low reactivity toward oxidationŌĆōreduction, and consistent behaviour across a wide range of chemical systems. The planar triangular geometry of COŌéā┬▓Ōü╗ also produces symmetrical charge distribution, which is a major reason carbonate ions strongly attract cations and form very stable ionic salts.
In sodium carbonate, the carbonate ion bonds ionically with two sodium ions, each carrying a +1 charge, balancing the 2ŌłÆ charge on the carbonate ion. In the solid state, NaŌééCOŌéā exists not as separate molecular units but as an extended ionic lattice, where NaŌü║ ions and COŌéā┬▓Ōü╗ ions arrange themselves in a repeating three-dimensional network governed by electrostatic attraction. This lattice structure gives sodium carbonate its crystalline form, relatively high melting point, and characteristic behaviour when heated. However, unlike many metal carbonate compounds that decompose easily when heated, sodium carbonate maintains structural integrity under high temperatures, a behaviour that has major implications for glass and ceramics manufacturing. The stability of the carbonate ion within this lattice explains why sodium carbonate is widely used industrially and why its decomposition is not trivial.
When dissolved in water, sodium carbonate behaves in a way that vividly illustrates the concepts of ionic dissociation and chemical equilibria. Water molecules attack the crystal lattice and break it apart, hydrating sodium ions and carbonate ions separately. The sodium ions disperse into solution, fully solvated and chemically unreactive except for maintaining electrical neutrality. The carbonate ion, however, immediately reacts with water in a series of equilibria that define its alkalinity. The carbonate ion acts as a base by accepting a proton from water and forming bicarbonate (HCOŌéāŌü╗), while releasing hydroxide (OHŌü╗) into the solution. This reaction accounts for the strongly alkaline nature of sodium carbonate solutions. At lower pH, bicarbonate may further convert to carbonic acid (HŌééCOŌéā), which decomposes into water and carbon dioxide. These interconnected pathways demonstrate that carbonate exists on an equilibrium ladder and that its basicity is directly controlled by its molecular structure, specifically its ability to accept protons while retaining resonance stabilization.
The buffering capacity of carbonateŌĆōbicarbonate solutions is one of sodium carbonateŌĆÖs most important chemical roles. Because the carbonate ion and bicarbonate ion exist in equilibrium, sodium carbonate can regulate pH by consuming hydrogen ions when acidity increases or by releasing hydrogen ions through the reverse reaction when alkalinity increases. This buffering mechanism is essential in both industrial and biological systems. Although sodium carbonate itself is too alkaline to function within living cells, the carbonateŌĆōbicarbonate system derived from it plays a crucial role in natural water bodies and atmospheric COŌéé exchange. In freshwater and marine chemistry, carbonate and bicarbonate ions regulate pH and influence the solubility of calcium salts, controlling coral and shell formation. These relationships highlight how the carbonate ion connects laboratory chemistry with planetary environmental systems.
Sodium carbonateŌĆÖs practical uses are deeply rooted in its structural chemistry. In laundry detergents, sodium carbonate acts as a water softener by binding calcium and magnesium ions that otherwise interfere with cleaning. The carbonate ion forms insoluble precipitates with these metal ions, removing them from the washing solution and allowing surfactants to function effectively. This process reflects the strong affinity between divalent metal ions and the doubly charged carbonate ion. In the production of glass, sodium carbonate lowers the melting temperature of silica by modifying the silicate network, reducing the energy needed to create liquid glass. In this process, carbonate decomposes at high temperature to release carbon dioxide and form sodium oxide, which then integrates into the silica lattice and increases fluidity. This behaviour shows how structural breakdown at high temperature supports new structural formation in materials.
Sodium carbonate is also a key reagent in acidŌĆōbase neutralization reactions. When added to an acid, it reacts vigorously, forming carbon dioxide and water while converting the acidŌĆÖs conjugate ions into corresponding salts. The reaction is driven by proton transfer from the acid to the carbonate ion and the intrinsic instability of carbonic acid, which decomposes rapidly. Beyond laboratory demonstrations, this neutralization property is widely used in industrial waste treatment to control acidity in effluents and in baking applications to regulate dough rise when combined with acidic components. In swimming pools and aquaponics systems, sodium carbonate is used to increase pH by neutralizing excessive hydrogen ions, bringing water chemistry back into balance.
The thermal behaviour of sodium carbonate reveals more structural relationships. Unlike many metal carbonates that decompose into metal oxides and carbon dioxide even at relatively low temperatures, sodium carbonate remains stable until extreme temperatures are reached. This unusual resistance is partly due to the large hydration energy released when sodium ions bond with carbonate, creating a system that favours the ionic assembly. The decomposition that eventually occurs produces sodium oxide and carbon dioxide, but only under intense heatŌĆöconditions that glassmaking industries exploit. When hydrated sodium carbonate (washing soda) is heated, it first undergoes dehydration, losing water without breaking the carbonate ion. This staged thermal transformation illustrates how structural water acted not as random moisture but as an integral part of the crystal lattice.
Despite being widely used in households, sodium carbonate requires responsible handling because of its alkalinity. In concentrated form or as dust, it can irritate skin, eyes and respiratory tissues because hydroxide ions released in moisture-rich environments attack sensitive biological molecules. However, in controlled concentrations, it is safe and plays a role in food processing, water treatment and pharmaceutical manufacturing. Environmentally, sodium carbonate breaks down into carbon dioxide and ions already common in nature, meaning it does not persist as a pollutant when used responsibly.
The educational value of sodium carbonate lies in the multitude of chemistry concepts that are visible through its structure and behaviour. The compound demonstrates ionic bonding, resonance in polyatomic ions, hydration of ions in solution, equilibrium between carbonate and bicarbonate ions, acidŌĆōbase reactivity, precipitation chemistry, buffering theory, thermal dehydration and the transformation of salts during high-temperature processes. Every macroscopic behaviourŌĆöwhether lowering the melting point of silica, neutralizing acids, softening water or stabilizing pHŌĆöcan be traced directly to the carbonate ionŌĆÖs resonance-stabilized planar geometry and its attraction to sodium ions in the lattice.
In summary, sodium carbonate is more than an ingredient in detergents or a raw material in manufacturing; it is a profound chemical model that showcases how atomic structure governs behaviour across the physical world. Its resonance-stabilized carbonate ion explains alkalinity, buffering, precipitation and environmental chemistry, while the sodium ions ensure structural balance and solubility without interfering in reactions. From laundry and glass manufacture to acidŌĆōbase equilibria and planetary carbon cycles, sodium carbonate proves that understanding the invisible organisation of electrons and charges is the key to explaining the visible behaviour of matter.