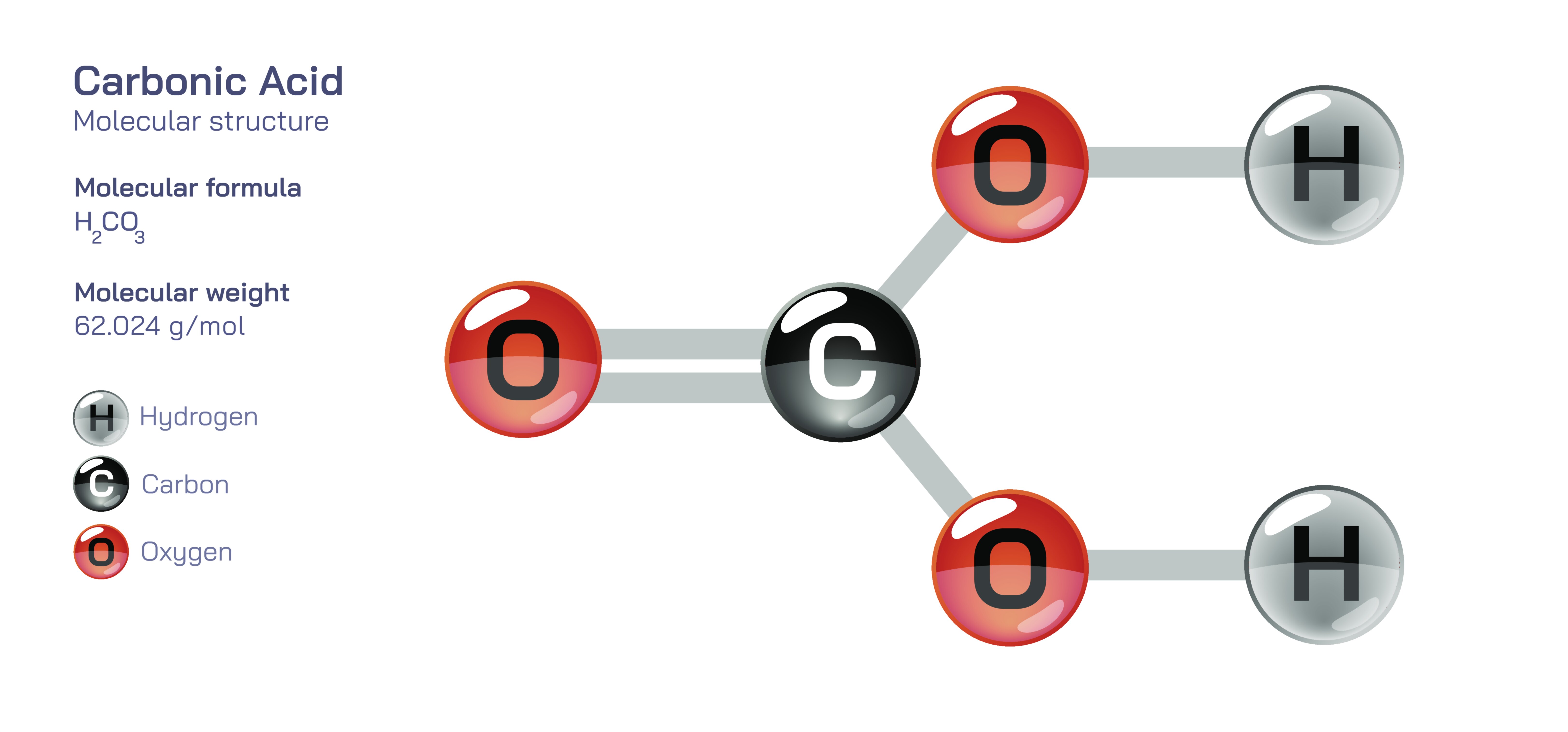
Carbonic Acid — Weak Acid Compound
Carbonic acid is one of the most essential yet understated compounds in natural chemistry, biology, environmental science, and planetary regulation. Although it is classified as a weak acid, its influence is anything but weak, because it stands at the center of countless processes that govern life, climate, water chemistry, and the balance of gases in the atmosphere. It is formed through a simple reaction—when carbon dioxide dissolves in water—but the consequences of that interaction shape entire ecosystems, support vital biochemical pathways, regulate blood pH in humans and animals, and influence the long-term geological transformation of Earth’s surface. To understand carbonic acid is to understand the subtle equilibrium that connects the atmosphere to the hydrosphere, the biosphere, and even the geosphere. Its molecular structure and behavior enable it to act as a bridge between gaseous carbon dioxide and ionic carbonate forms, making it one of the most important intermediates in Earth’s carbon cycle.
Molecularly, carbonic acid forms when carbon dioxide interacts with water molecules, creating a hydrated compound that exists only fleetingly before dissociating or reverting back to carbon dioxide and water. It is not a stable acid in the way more familiar mineral acids are; instead, it exists continuously in dynamic equilibrium. The structure consists of a central carbon atom surrounded by oxygen atoms, with hydrogen atoms bonded in a way that allows the molecule to donate protons under appropriate conditions. Because of this configuration, carbonic acid can behave as a proton donor, contributing to acidity, yet it releases its hydrogen ions only partially, which is why it is classified as a weak acid. Its weak dissociation is precisely what makes it invaluable in natural buffering systems, because it prevents sudden drastic changes in pH that could harm living organisms or destabilize chemical environments.
In the human body, carbonic acid plays a crucial role in maintaining acid–base balance, a delicate equilibrium that keeps blood pH within a narrow range compatible with life. When cells produce carbon dioxide during respiration, the gas diffuses into the bloodstream, where it reacts with water to form carbonic acid. This acid then partially dissociates into bicarbonate and hydrogen ions, forming a buffering pair that stabilizes the blood’s pH. Specialized enzymes in red blood cells accelerate this interconversion, allowing the body to regulate acidity in real time as metabolic demands change. During exhalation, the equilibrium shifts again: carbonic acid converts back into carbon dioxide, which is released from the lungs. This reversible transformation is one of the most elegant physiological systems humans possess. Without the constant interplay between carbon dioxide, carbonic acid, and bicarbonate, maintaining consistent blood chemistry would be impossible, and even slight metabolic shifts could have life-threatening consequences.
In natural waters—streams, lakes, oceans, and underground aquifers—carbonic acid shapes the chemistry that supports aquatic ecosystems. Rainwater naturally absorbs carbon dioxide from the atmosphere, forming weak carbonic acid that contributes to its slight acidity. This acidic rainwater seeps into soils and dissolves minerals, carrying essential nutrients to plants and microorganisms. In larger bodies of water, carbonic acid influences the balance between dissolved carbon dioxide, bicarbonate ions, and carbonate ions. This balance affects the solubility of many minerals, the buffering capacity of water, and the ability of aquatic organisms to form shells and skeletons. Marine life such as corals, mollusks, and plankton depend on carbonate ions to build calcium carbonate structures. However, when carbon dioxide levels increase in the atmosphere, more of it dissolves in the oceans, forming additional carbonic acid that shifts the equilibrium toward bicarbonate and reduces carbonate availability. This shift drives ocean acidification, a phenomenon that threatens marine ecosystems by weakening coral reefs, impairing shell formation, and destabilizing food chains. Carbonic acid therefore acts as both a natural regulator and a sensitive indicator of environmental changes.
Carbonic acid also plays a central role in geological processes that shape the Earth’s surface. When carbonic acid in rainwater interacts with rocks—especially those containing calcium carbonate such as limestone—it begins a slow process of chemical weathering. This reaction dissolves the rock, carrying dissolved minerals into rivers and eventually to the ocean. Over long periods, this process contributes to the creation of caves, sinkholes, and karst landscapes, demonstrating how a seemingly weak acid can sculpt entire geological structures. At the same time, dissolved minerals transported by carbonic acid eventually precipitate out in marine environments, forming sedimentary rocks that lock carbon away for millions of years. This interplay forms part of Earth’s long-term carbon cycle, helping regulate global carbon dioxide levels and contributing to climate stability over geological timescales.
In industrial contexts, carbonic acid appears in numerous applications, even though it is rarely handled directly in its pure form. It is the active agent responsible for the fizz and tanginess in carbonated beverages. When pressurized carbon dioxide dissolves in water, carbonic acid forms, giving soft drinks their characteristic bite and mild acidity. In cleaning and sanitation products, carbonic acid arises when carbon dioxide is released into water-based solutions, helping remove deposits or residue without the harsh effects of stronger acids. In environmental engineering and water treatment, the carbonic acid system helps adjust pH, remove impurities, and maintain balanced water chemistry. Its ability to shift smoothly between dissolved carbon dioxide, carbonic acid, and bicarbonate makes it one of the most versatile tools for managing pH in both natural and engineered systems.
In plant physiology, carbonic acid serves as an intermediate in the uptake and utilization of carbon dioxide during photosynthesis. Although plants absorb carbon dioxide directly rather than carbonic acid, the dissolved form in water droplets on leaves or in plant tissues influences stomatal behavior, hydration levels, and the microenvironment around plant cells. In soils, carbonic acid contributes to nutrient cycling by dissolving mineral particles and releasing essential ions such as calcium, magnesium, and potassium. These nutrients become available for plant absorption, supporting growth and overall soil fertility. Thus, carbonic acid acts not only as a chemical intermediary but also as a driver of ecosystem productivity.
From a physical chemistry perspective, carbonic acid’s behavior is defined by its equilibrium with carbon dioxide and bicarbonate. These reversible transformations demonstrate principles of chemical equilibrium, gas solubility, and acid–base regulation. Because carbonic acid forms and decomposes rapidly, it allows systems such as oceans and blood plasma to respond to environmental changes almost instantly. This responsiveness makes carbonic acid one of the most important components of natural buffering systems, ensuring that pH remains within stable, life-supporting limits even when external conditions fluctuate dramatically.
As climate change reshapes global systems, carbonic acid’s role becomes even more central. Rising carbon dioxide levels increase carbonic acid formation in oceans, altering chemical balances that have remained stable for centuries. These changes highlight how interconnected the world’s systems are: human activities influence atmospheric composition, which alters carbonic acid levels, which in turn affect ocean chemistry, biological health, and geological processes. Understanding carbonic acid is therefore essential not only for chemists but for environmental scientists, medical researchers, geologists, and policymakers.
Ultimately, carbonic acid is a reminder that the most important compounds are often the ones that work quietly in the background. It possesses no dramatic strength or volatile characteristics; instead, its power lies in its equilibrium behavior, its role as a mediator, and its ability to connect diverse systems through a single chemical framework. From the breath we exhale to the oceans that cover most of the planet, from the rocks beneath our feet to the beverages we drink, carbonic acid shapes the world in ways both subtle and profound. Understanding it means understanding some of the most fundamental processes that sustain life and regulate Earth’s dynamic environment.
Carbonic Acid — Weak Acid Compound Vector Illustration
Carbonic acid vector illustration showing molecular structure and formation from carbon dioxide and water for chemistry and educational use.
Summary
File Details
File Type
-
EPS
File Size
-
8.28 MB
File Dimension
-
Scalable vector file
Your contribution helps us create more resources, but please support only if it's comfortable and affordable for you.❤️️