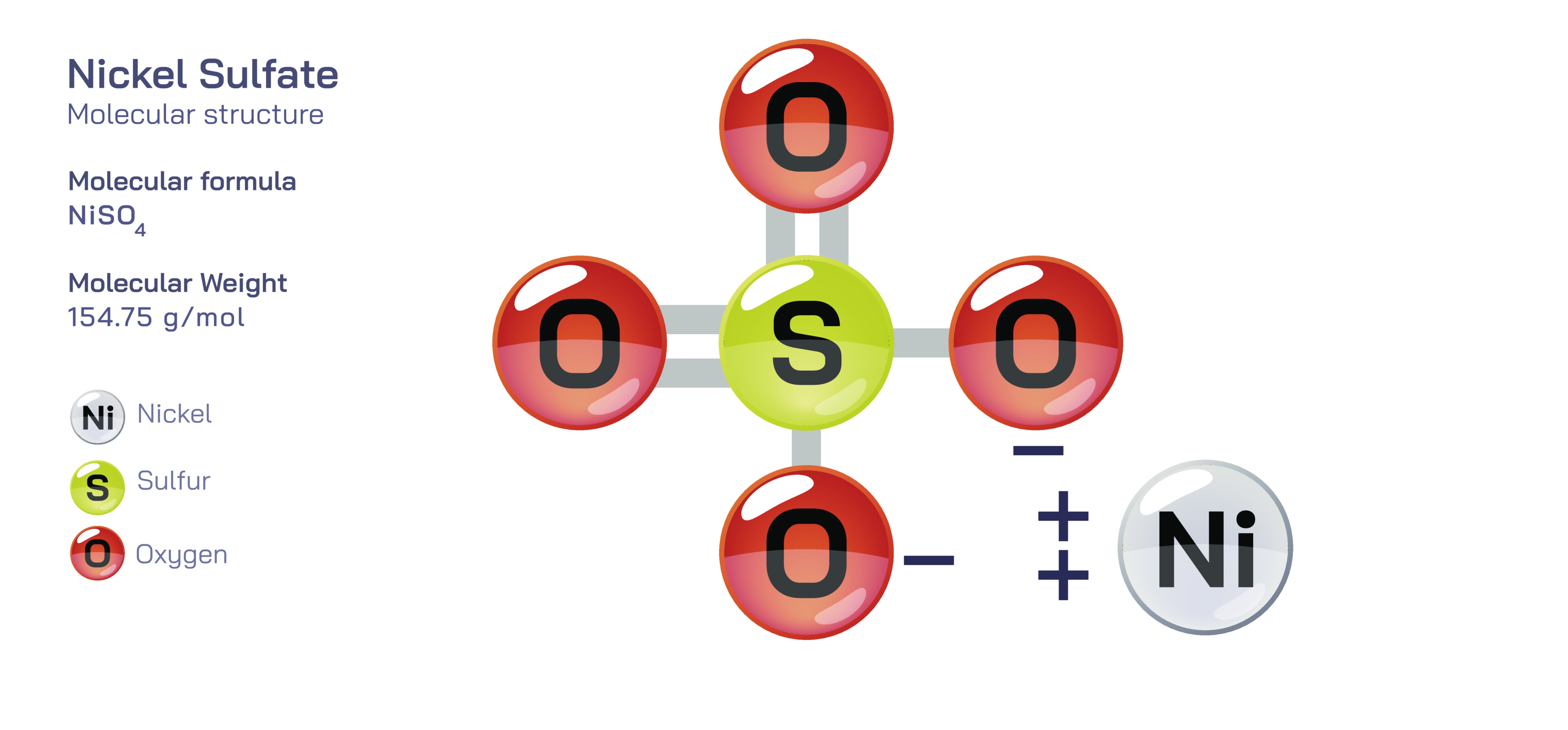
Nickel Sulfate — Structure, Properties, and Industrial Uses
Nickel sulfate is an inorganic compound whose molecular structure and physicochemical identity have enabled it to become one of the most important materials in metal finishing, battery manufacturing, catalyst synthesis, electroplating, pigments, advanced ceramics, and chemical processing. Commonly encountered as nickel(II) sulfate hexahydrate with the formula NiSO₄·6H₂O, the compound consists of a divalent nickel cation bonded electrostatically to a sulfate anion, and in the hydrated crystalline form, six water molecules coordinate within the lattice to stabilize the complex. The sulfate anion is a tetrahedral group containing a central sulfur atom surrounded by four oxygen atoms with symmetrical resonance patterns that distribute electron density evenly across the oxygens, and in solution this anion dissociates from the nickel cation to create a highly ionic aqueous environment in which Ni²⁺ remains solvated by water molecules. When represented in a vector illustration, nickel sulfate appears as a central nickel ion surrounded in octahedral geometry by water molecules, accompanied by a sulfate group visualized as a tetrahedral arrangement of oxygen atoms around sulfur. This type of illustration helps highlight that the nickel center maintains high reactivity and mobility even within the hydrated structure, which explains why the compound dissolves rapidly, maintains strong ionic conductivity, and delivers nickel ions with great efficiency into electrochemical, catalytic, and industrial systems. The coordination chemistry of nickel sulfate reflects its ability to serve as both a stable storage form and an active participant in technologically important reactions, demonstrating how the microscopic structure of matter governs large-scale industrial behavior.
The most significant application of nickel sulfate globally is its use in electroplating, especially in the formation of durable, corrosion-resistant nickel coatings on metals. In electroplating solutions, nickel sulfate provides a controlled and continuous source of Ni²⁺ ions that migrate toward the cathode under an applied electric current, depositing as metallic nickel on surfaces such as steel, copper, brass, and aluminum. The sulfate anion enhances conductivity and ensures uniform ionic distribution throughout the bath, while the hydrated nickel structure maintains predictable solubility and avoids premature precipitation. This process creates nickel layers that improve wear resistance, impact protection, thermal stability, corrosion resistance, and aesthetic finish on industrial components ranging from automotive parts and consumer appliances to aerospace fittings, marine equipment, and precision hardware. In decorative and engineering nickel plating, nickel sulfate remains indispensable because the coating quality depends on the ability to maintain steady nickel ion activity, predictable pH, current distribution, and deposition kinetics. The molecular structure of nickel sulfate supports these requirements by providing strong interactions with solvents that maintain bath homogeneity and, in alloy plating with materials such as cobalt or iron, nickel sulfate becomes a key precursor to multimetal coatings with tailored hardness, luster, and mechanical performance.
Nickel sulfate also plays a critical role in modern energy technology because it has become a foundational raw material in the production of nickel-rich cathode formulations for lithium-ion batteries. Cathode chemistries such as NMC (nickel–manganese–cobalt) and NCA (nickel–cobalt–aluminum) depend on high-purity nickel sulfate as the precursor for controlled precipitation of layered metal hydroxides that are later calcined to form electrochemically active oxides. Nickel contributes greatly to energy density by supporting stable reversible redox reactions, which is why advanced electric vehicle batteries increasingly use cathodes with high nickel content. The transformation from nickel sulfate solution to complex battery oxide relies on precise control of stoichiometry, impurity levels, particle morphology, and hydration state, and the ability of nickel sulfate to dissolve uniformly and precipitate cleanly makes it the preferred industrial starting compound. As the global transition to electric mobility accelerates, large-scale manufacturing of nickel sulfate has expanded because every metric ton of nickel-rich cathode material begins as a controlled chemical synthesis in which nickel sulfate supplies the nickel ions that become the backbone of high-capacity energy storage. The compound therefore forms an invisible but essential bridge between the extraction of raw nickel and the development of next-generation sustainable energy infrastructure.
In addition to its involvement in electroplating and energy storage, nickel sulfate plays a transformative role in catalysis and materials chemistry. Upon thermal decomposition or controlled precipitation, nickel sulfate becomes a route to producing nickel oxide, mixed-metal oxides, and structured catalytic materials that support hydrogenation, methane reforming, water splitting, carbon dioxide reduction, hydrodesulfurization, ammonia cracking, and numerous organic transformation reactions. The sulfate group breaks down during heating without leaving carbon-based residues, allowing nickel to integrate cleanly into ceramic frameworks and catalytic substrates. In industrial catalytic converters, nickel oxide derived from nickel sulfate can assist in breaking down pollutants or enhancing the reactivity of reforming catalysts. In laboratory synthesis, nickel sulfate is widely used as a precursor for complexes containing organic ligands, and its solubility facilitates ligand-exchange reactions that create tailored organometallic catalysts used in fine chemical synthesis, polymer formation, and pharmaceutical intermediates. The compound’s hydration sphere enables it to transition between different coordination environments during synthesis, meaning its molecular structure does not trap nickel in a rigid configuration but rather allows it to reorganize under chemical or thermal influence.
Nickel sulfate is also indispensable in ceramic and glass coloration, where thermal decomposition introduces nickel into silica-based or oxide-based materials to create color effects ranging from greys and greens to deep blues depending on the firing atmosphere and co-dopants. Because nickel sulfate disperses uniformly in ceramic slurries and melts before decomposition, the resulting nickel-containing oxide phases distribute evenly through the material, preventing streaking or uneven coloration in glazes and fired products. Nickel-containing ceramic materials created using nickel sulfate display enhanced mechanical strength, thermal durability, and magnetic or dielectric properties, making the compound valuable in technical ceramics that support electronic components, sensors, ferrites, and high-temperature structural applications. Pigment manufacturing also utilizes nickel sulfate for complex color compounds used in industrial paints, specialty coatings, and plastics where stability under UV light and heat cycling is essential.
In environmental science and wastewater treatment research, nickel sulfate is frequently used not as a treatment agent but as a controlled source of nickel in studies of ion exchange, heavy metal adsorption, membrane filtration, and catalytic water purification. Because the compound dissolves completely and predictably, it allows engineers and researchers to examine how filtration media, biopolymers, activated carbon, and metal-oxide composites capture and immobilize nickel ions. It also provides the nickel component for catalysts that degrade industrial dyes, organic pollutants, and pharmaceutical residues through photocatalysis, electro-oxidation, or redox reactions. Furthermore, in agriculture and microbiology research, trace nickel from nickel sulfate may be used to examine biological systems that depend on nickel-containing enzymes, although this application must be controlled carefully because nickel is an essential micronutrient only in very small quantities and becomes harmful in excess.
The physical properties of nickel sulfate, such as its crystalline structure, solubility, and thermal behavior, relate directly to the coordination chemistry of the nickel center and the sulfate group. The hexahydrate melts in its own hydration water and dehydrates stepwise when heated, eventually forming anhydrous nickel sulfate and then nickel oxide as temperature rises further. This multi-stage transformation provides predictable thermal pathways that are critical in ceramics and catalyst production. The bright green crystals of the hydrated compound, the high ionic conductivity of aqueous solutions, the stability of the sulfate group in acidic environments, and the reactivity of nickel ions in ligand exchange illustrate how macroscopic industrial attributes emerge from microscopic structural design. Each property — from solubility to decomposition — becomes a functional lever that engineers and chemists use to fabricate materials and coatings with predictable performance and long-term stability.
Despite its widespread industrial importance, nickel sulfate requires careful and responsible handling because nickel compounds can trigger allergic skin sensitization and can exhibit toxicity in aquatic environments when unregulated releases occur. Occupational exposure must be controlled through ventilation, protective equipment, and safe chemical handling procedures, while liquid wastes containing nickel ions are typically treated through precipitation, ion exchange, or electrochemical recovery processes to prevent contamination and enable recycling of valuable nickel. Electroplating facilities and battery-material production plants frequently implement closed-loop recycling systems to recapture nickel sulfate from spent solutions, reducing environmental burden while supporting economic efficiency. Safety management does not diminish the significance of nickel sulfate in industry; rather, it reflects an awareness that powerful and beneficial materials must be managed with respect for human health and environmental responsibility.
Across all of its applications — electroplating, anodizing sealants, nickel-rich battery cathodes, catalytic materials, pigments, ceramics, and environmental technology — nickel sulfate demonstrates how a compound’s industrial usefulness depends directly on its molecular structure. The octahedral hydration of the nickel center, the stability and charge distribution of the sulfate group, the solubility profile of the crystalline salt, and the thermal decomposition pathway together define how this material behaves during manufacturing and how it becomes embodied in final products. Nickel sulfate illustrates the profound truth that the architecture of atoms governs the functionality of technology at every scale. From the plated bolts that hold aircraft panels together, to the cathode in an electric vehicle battery storing energy for transportation, to catalysts cleaning industrial emissions, to pigments that color durable ceramic tiles, a simple crystalline compound becomes a critical part of modern engineering because the structure of matter determines the structure of industry.