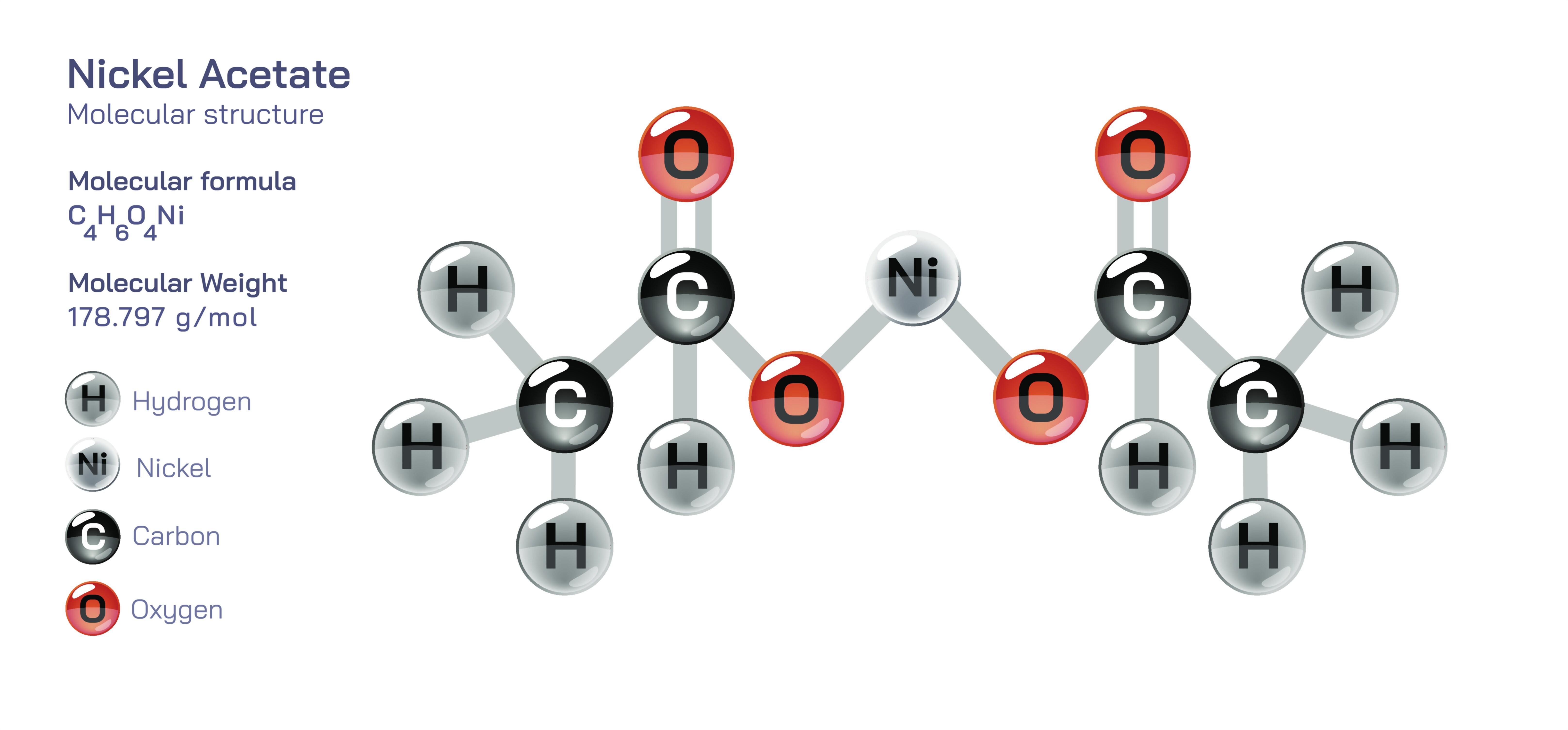
Nickel Acetate — Chemical Compound Structure and Industrial Uses
Nickel acetate is a coordination compound in which nickel ions are bonded with acetate ligands, and its molecular structure is the reason it plays such an important role in electroplating, surface finishing, materials science, catalytic synthesis, ceramics, textile processing, and numerous industrial chemical systems. At its core, nickel acetate contains a divalent nickel cation (Ni²⁺) associated with two acetate anions derived from acetic acid, forming a stable chelated complex in which the oxygen atoms of the carboxylate groups act as electron-pair donors that coordinate to the nickel center. In its most common commercial and laboratory form, nickel acetate is encountered as nickel(II) acetate tetrahydrate, a crystalline green compound in which four water molecules integrate into the coordination sphere without altering the essential nickel–acetate bonding network. This structure explains its solubility in water and polar solvents, its moderate thermal stability, and its predictable dissociation behavior. When a vector illustration of nickel acetate is produced, the acetate groups appear as functional units containing a methyl group linked to a carboxylate moiety, while the nickel ion sits at the center of an octahedral coordination field surrounded by oxygen donors from acetate and water molecules. This visual representation captures the fundamental concept that the nickel ion remains accessible within a coordination environment, which determines how the compound behaves when dissolved, heated, or used in reactions that involve ligand exchange or controlled deposition of nickel.
One of the most important industrial applications of nickel acetate is in electroplating and metal finishing, where controlled deposition of nickel onto metal surfaces protects components from corrosion, wear, and impact damage. Nickel acetate dissolves readily in plating baths to release Ni²⁺ ions, which migrate under applied electric current and form metallic nickel layers on substrates such as steel, copper, brass, and aluminum. The acetate ligands play a stabilizing role by preventing premature precipitation of nickel salts and helping maintain ideal bath chemistry under fluctuations of pH, heat, or current density. In anodized aluminum finishing — especially in aerospace, automotive, and architectural metal systems — nickel acetate is used in sealing baths that form protective complexes inside microscopic pores created during anodization. This sealing process increases resistance to moisture, abrasion, and chemical attack, preserves dye coloration on anodized aluminum, and extends the service life of components exposed to harsh environmental conditions. Here the structural characteristics of nickel acetate make a direct contribution: the nickel ion reacts with the hydroxyl-rich oxide layer on aluminum to create a sealed structure that reduces permeability and improves surface integrity, while the acetate groups ensure controlled solubility and diffusion through the sealing bath. Without the specific coordination behavior of nickel acetate, this treatment would be far less effective, demonstrating how industrial functionality begins at the molecular level.
Nickel acetate also plays a major role in the synthesis of catalysts and advanced materials because the nickel atom at the center of the compound can be used to construct a wide variety of nickel-based chemical frameworks. When nickel acetate is heated or treated with other ligands, the acetate groups may be displaced, allowing for the formation of new nickel complexes that possess different catalytic or electronic properties. Researchers use nickel acetate as a precursor when producing nickel nanoparticles, metal–organic frameworks, organometallic catalysts, battery materials, and composite coatings because it offers a controlled and predictable pathway for delivering nickel ions into chemical systems. In polymer chemistry, it functions as a co-catalyst in reactions requiring metal activation, and in research laboratories it serves as a reagent for assembling ligated complexes with tailored magnetic or redox behavior. The reason for this versatility lies in the geometry of the nickel coordination sphere: the acetate ligands bind strongly enough to stabilize the metal in storage and handling, but weakly enough to allow ligand exchange under reaction conditions. When scientists design new materials for hydrogenation catalysis, renewable energy processes, organic transformation reactions, or electronic component fabrication, nickel acetate often becomes the chosen starting point because it combines ease of handling with structural accessibility and reliable catalytic transformation.
Ceramic and glass manufacturing also depend on nickel acetate because it becomes a convenient route for introducing nickel oxide into high-temperature materials. When nickel acetate is subjected to calcination or fired inside ceramic glazes, the acetate ligands thermally decompose while the nickel core oxidizes and integrates into the ceramic matrix. Nickel oxide produced in this way contributes attributes such as color, hardness, thermal resistance, and structural stability. In enamel coatings and glass coloration, nickel compounds derived from nickel acetate influence the tonal range of pigments and modify optical characteristics. Nickel acetate is particularly valued in these applications because it disperses uniformly in the glazing mixture before decomposition, preventing uneven color distribution and unpredictable crystallization behavior. In nickel ferrite and other functional ceramic materials used in magnets and electronic components, nickel acetate serves as a precursor during sintering processes that require precise chemical composition and controlled cation distribution across the lattice. These material transformations — from green crystalline nickel acetate to durable nickel-infused ceramic structures — reflect how chemical identity expressed at room temperature becomes an engineering property when subjected to heat and integration with other inorganic elements.
In the field of inks, pigments, and textile dyeing, the coordination chemistry of nickel acetate enables it to act as a mordant — a compound that stabilizes dyes on fibers and improves wash resistance, color depth, and durability. Nickel ions form coordination complexes with dye molecules and binding sites on fabric surfaces, helping pigments resist fading and improving the visual richness of printed patterns. In specialized ink systems used on ceramics, plastics, metals, and textiles, nickel acetate provides enhanced adhesion properties through the formation of metal–dye complexes that remain stable during drying, firing, or exposure to water and light. These interactions highlight that the industrial usefulness of nickel acetate stems not from the acetate ligand alone but from the ability of the nickel ion to anchor chromophores and bond with functional groups present in commercial dyes and polymeric binders.
Nickel acetate even plays a role in environmental science and water treatment research by acting as a controlled source of nickel ions in experiments focused on metal sorption, photochemical degradation of pollutants, and catalyst-driven breakdown of organic contaminants. In electrochemical studies, it is used to prepare nickel-based electrodes and nickel oxide catalysts that support reactions relevant to hydrogen evolution, CO₂ reduction, wastewater remediation, and energy storage. Because the acetate groups affect electron density around the nickel center and influence the morphology of nickel-derived materials, the compound enables researchers to tailor surface area, catalytic activity, and conductivity in engineered materials.
Like all nickel compounds, nickel acetate must be handled responsibly because nickel exposure can cause allergic sensitization and environmental toxicity. Industrial users limit airborne dust, prevent contamination of water systems, and incorporate ventilation, protective equipment, and recovery methods to reclaim nickel from waste solutions. These measures do not diminish the value of the compound; they reflect respect for chemical safety and environmental stewardship in industries that depend on nickel chemistry for protective coatings, corrosion control, and advanced material fabrication.
Across all of its applications, nickel acetate demonstrates a consistent principle: the functional value of a chemical compound begins with its structure. Its coordination geometry stabilizes nickel in solution, enables precise ion release in electrochemical systems, supports ligand exchange in catalytic synthesis, promotes uniform decomposition into oxides at high temperatures, and bonds to dyes and substrates in pigment systems. Whether it seals the pores of anodized aluminum, forms the basis of a high-performance catalyst, contributes to ceramic materials, or enhances the durability of textile pigments, nickel acetate translates microscopic molecular architecture into macroscopic industrial performance. The compound highlights how the science of coordination chemistry extends far beyond the laboratory and into manufacturing plants, energy research, material development, surface engineering, and protective finishing — all because of the way one nickel ion and two acetate groups arrange themselves to create a structure capable of supporting some of the most important technologies used across modern industries.