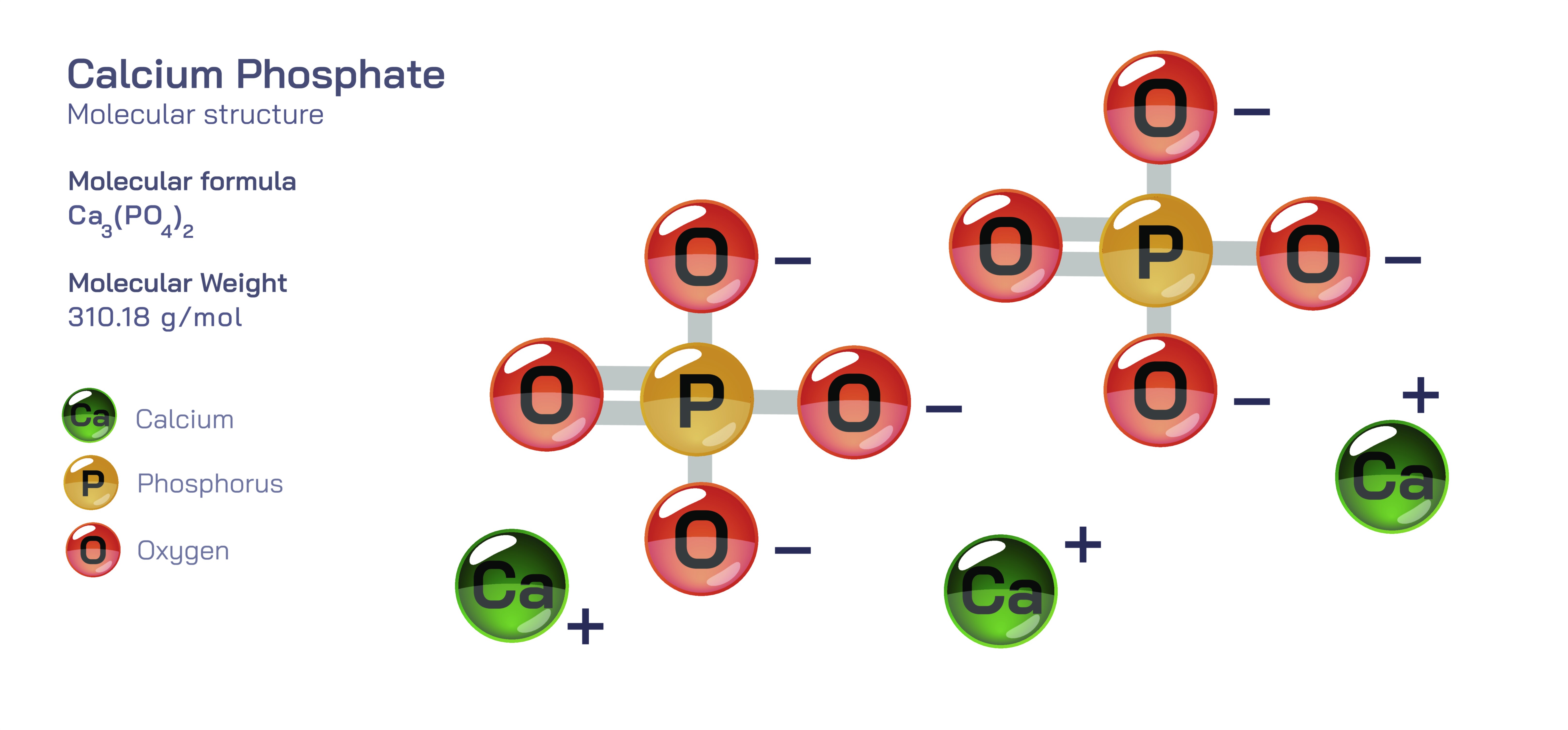
Calcium Phosphate — Inorganic Mineral Compound
Calcium phosphate is an exceptionally important inorganic mineral compound whose presence spans biological systems, geological formations, industrial applications, and essential processes that support life on Earth. Its significance becomes clear when considering how deeply embedded it is in the structure and function of organisms, particularly in vertebrates, where it forms the backbone—both literally and figuratively—of skeletal development. Even without resorting to formulas or mathematical descriptions, calcium phosphate can be understood as a family of compounds in which calcium ions interact with phosphate ions to create solid, stable, mineral-like structures. These interactions produce hard, durable frameworks that resist compression, maintain shape, and support the forces encountered by biological systems. When these minerals accumulate in organized patterns such as those found in bones and teeth, they transform soft tissues into rigid but dynamic structures that enable movement, protection, strength, and long-term survival. The concept behind calcium phosphate extends beyond biology, reaching into environmental chemistry, industrial manufacturing, food science, agriculture, and medicine. Its versatility arises from the way calcium and phosphate work together to generate forms that are simultaneously stable and reactive depending on their conditions.
At the center of its biological relevance is its role in bones and teeth, where specialized forms of calcium phosphate create an intricate solid matrix. This structure provides the mechanical properties that define the skeletal system. Bones are not simply static frameworks; they are metabolically active tissues that constantly undergo renewal, micro-repair, and remodeling. Calcium phosphate crystals embed themselves within a matrix of collagen fibers, forming a composite material that combines rigidity with a degree of resilience. This balance allows bones to withstand stress while also absorbing impact. Teeth rely even more heavily on specialized forms of calcium phosphate, particularly in enamel, which represents the hardest natural substance in the human body. Enamel’s strength arises from an extremely dense, highly ordered mineral arrangement that resists decay, mechanical wear, and chemical erosion. Without the dependable mineralization provided by calcium phosphate, vertebrate life would not have evolved the structural diversity that allows for locomotion, feeding, defense, and environmental interaction.
The formation and maintenance of calcium phosphate structures in the body depend on delicate physiological processes that involve hormones, blood chemistry, diet, and cellular regulation. The body constantly maintains a careful equilibrium between dissolved calcium, phosphate, and the mineral forms stored in bones. This balance enables mineral mobilization when needed—such as during growth, pregnancy, lactation, or injury repair—while preventing excessive accumulation that could cause calcification in soft tissues. Specialized cells such as osteoblasts and osteoclasts orchestrate the deposition and resorption of calcium phosphate within bone tissues, maintaining skeletal strength while regulating the supply of essential ions throughout the body. These processes highlight how calcium phosphate is not merely a substance but part of a dynamic system essential to life. Its solubility, structural stability, and capacity to form different mineral phases enable biological systems to build, dissolve, reshape, and reorganize skeletal components in response to physiological needs.
In environmental chemistry, calcium phosphate plays an equally critical role, especially in soil composition and nutrient cycling. Phosphate is a limiting nutrient in many ecosystems because plants require it for nucleic acid formation, energy transfer, and metabolic regulation. However, phosphate often binds with calcium in soils, creating mineral deposits that are not immediately accessible to plant roots. The slow, natural weathering of calcium phosphate minerals releases phosphate over long periods, enabling ecosystems to flourish through sustained nutrient availability. This long-term nutrient management is one of the reasons why phosphate-rich sediments and rocks become foundational components of agricultural fertilizers. When these minerals are mined and processed, they re-enter the biological cycle through crop fertilization, supporting global food production systems. The cycling of calcium phosphate through geological and biological pathways illustrates the interconnectedness of soil chemistry, plant life, and human agriculture.
Industrially, calcium phosphate finds numerous uses that arise from its chemical stability, biocompatibility, and ability to act as a source of both calcium and phosphorus. One of its most important industrial applications occurs in the production of fertilizers, where phosphate rock is processed to extract phosphorus compounds for use in agriculture. This process supports high-yield crop production in modern farming systems. In another context, calcium phosphate appears in ceramics and biomaterials used in medical implants. Because its composition is closely related to that of natural bone, synthetic calcium phosphate materials integrate well with biological tissues, making them ideal for bone grafts, dental implants, and orthopedic reconstruction. These materials support natural bone regeneration, contributing to medical practices that restore skeletal function after trauma, disease, or surgery.
In food science, calcium phosphate serves multiple purposes, ranging from nutritional supplementation to functional food processing. It often appears as a source of dietary calcium in fortified products, including cereals, beverages, and nutritional supplements. In baking and food preservation, certain forms of calcium phosphate act as leavening agents, buffering compounds, or stabilizers that maintain product texture and quality. The compound’s inertness and safety make it suitable for widespread use in consumable goods. Its nutritional value derives from calcium’s essential role in bone health, nerve function, muscle contraction, and metabolic activity, while phosphate contributes to energy processing, cell membrane integrity, and genetic material structure.
Calcium phosphate also figures prominently in water treatment, industrial chemistry, and materials engineering. Its precipitation properties allow it to be used in processes that remove unwanted ions from water, contributing to purification and mineral balance. In materials science, researchers examine the structural properties of calcium phosphate to design biomimetic materials that simulate natural bone or enamel, exploring applications in regenerative medicine and advanced prosthetics. Its ability to form microstructured and nanostructured phases makes it a valuable subject in research focused on improving medical implants, controlled drug delivery, and surface coatings that enhance biocompatibility.
Culturally and historically, the presence of calcium phosphate has shaped human development through its role in bone artifacts, tools, ancient remains, and archaeological records. The durability of bone, driven largely by calcium phosphate content, enables scientists to study past civilizations, extinct species, and evolutionary processes through preserved skeletal remains. These remains hold stories of ancient diets, migration, lifestyles, and environmental conditions, offering insights that would not be possible without the stability of mineralized structures.
The versatility of calcium phosphate extends into cutting-edge research that explores how its properties can support emerging technologies. Scientists investigate its capacity for binding pharmaceuticals, delivering therapeutic agents, and supporting tissue engineering scaffolds designed to promote cellular growth. Its natural biocompatibility makes it an attractive candidate for developing the next generation of medical treatments and regenerative therapies. In nanotechnology, calcium phosphate nanoparticles show promise in imaging, gene delivery, and minimally invasive medical diagnostics, offering avenues for precision medicine.
Ultimately, calcium phosphate functions as a bridge between the geological world, the biological world, and the technological world. It provides the rigid structure that supports life, the nutrient cycle that sustains ecosystems, the industrial raw material that powers agriculture, and the biomaterial that advances modern medicine. Understanding calcium phosphate means recognizing its role in every level of natural and engineered systems, from microscopic cellular processes to massive geological cycles. It represents the harmony between minerals and living organisms, highlighting the profound influence that inorganic compounds have on the shape, function, evolution, and continuity of life.