Benzoic Acid — Understanding the Structure and Behavior of an Aromatic Carboxylic Compound
The Fundamental Identity of Benzoic Acid in Organic Chemistry
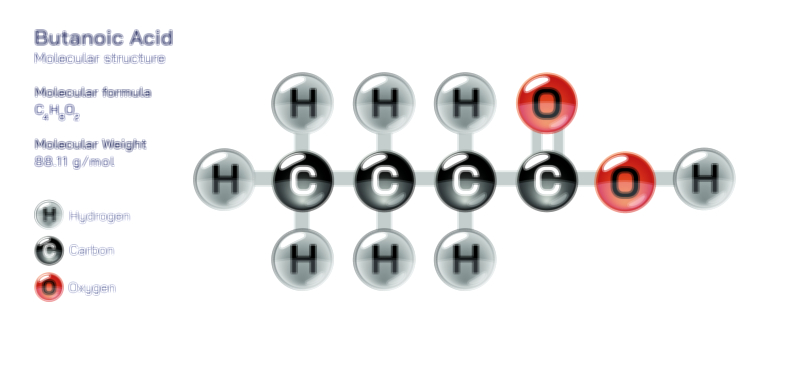
Benzoic acid is one of the most important aromatic carboxylic compounds, known for its simple yet influential structure and its presence across chemical, biological, and industrial applications. It consists of a benzene ring bonded to a carboxyl group, creating a molecule that blends the stability of an aromatic ring with the reactivity of an acidic functional group. This combination makes benzoic acid an excellent model for understanding how organic compounds behave when aromatic stability meets polarity and acidity. Recognized as a solid crystalline substance with a slightly sharp odor, benzoic acid appears naturally in plants, resins, and certain fruits. Its widespread presence and stability have made it an essential compound in preservation, synthesis, and material processing, demonstrating how modest chemical structures can have significant practical impact.
How the Aromatic Ring Shapes the Nature of Benzoic Acid
At the heart of benzoic acid lies a benzene ring, a structure known for its remarkable stability and symmetry. This ring anchors the molecule and protects it from rapid breakdown, allowing benzoic acid to maintain its integrity across a wide range of temperatures and environments. The aromatic ring is rich in electrons and forms a stable platform for chemical reactions. Although it does not easily undergo transformations that break its core structure, it participates readily in substitution reactions where various atoms or groups replace hydrogen atoms around the ring. This unique balance of resilience and flexibility makes benzene-based compounds central to organic chemistry, and benzoic acid is no exception. The ring not only stabilizes the molecule but also influences how the acidic group interacts with surrounding materials, contributing to the compound’s distinct behavior.
The Role of the Carboxyl Group in Determining Reactivity
Connected to the aromatic ring is the carboxyl group, the portion of the molecule responsible for benzoic acid’s acidic nature. This group draws electron density toward itself, giving the molecule a polar character and enabling it to interact strongly with water, alcohols, and other polar substances. The acidic behavior of benzoic acid is central to its role in preservation, synthesis, and chemical reactivity. In solid form, the molecules often arrange themselves in orderly crystalline patterns held together by strong interactions between carboxyl groups. These interactions give benzoic acid its characteristic melting behavior and help maintain its purity and stability during storage. The carboxyl group also acts as a reactive site in organic synthesis, allowing chemists to convert benzoic acid into countless derivatives through controlled transformations.
Solubility Behavior and Interaction with Different Environments
Benzoic acid demonstrates a characteristic solubility pattern that reflects its dual nature as both aromatic and acidic. It dissolves readily in hot water but only sparingly in cold water, a behavior linked to the stability of its crystalline form and the interactions between its molecules. In organic solvents, however, benzoic acid shows much greater solubility, especially in those that can interact with its aromatic ring or other polar groups. This distinctive solubility behavior helps manufacturers purify benzoic acid through crystallization and allows scientists to control its reactions with precision. In biological or natural environments, its limited solubility prevents it from dispersing too quickly, a feature that contributes to its effectiveness as a preservative by slowing the growth of microorganisms.
Natural Occurrence and Biological Relevance
Although simple in structure, benzoic acid occurs widely in the natural world. Plants produce it as part of their metabolic processes, and it appears in fruits such as cranberries, plums, and certain berries. In these natural settings, benzoic acid serves as a protective compound that helps plants defend themselves against harmful microorganisms. Its antimicrobial properties, derived from the acidic nature of its carboxyl group, inhibit the growth of bacteria and fungi under specific conditions. When incorporated into food preservatives, this same behavior helps prevent spoilage and extends the lifespan of perishable products. The biological relevance of benzoic acid shows how even small organic molecules can contribute to natural defense, metabolism, and ecological balance.
Industrial Uses in Preservation, Synthesis, and Materials
Benzoic acid plays a central role in many industrial processes due to its stability, reactivity, and antimicrobial properties. In the food industry, it serves as a foundational preservative in its salt forms, where it slows the growth of microorganisms in acidic foods and beverages. In chemical manufacturing, benzoic acid acts as a key starting material for producing dyes, plastics, synthetic fibers, and resins. Its reactive carboxyl group allows manufacturers to convert it into esters, amides, and other derivatives that appear in products ranging from perfumes to polymers. In pharmaceuticals, benzoic acid functions both as an ingredient and as a precursor to more complex therapeutic compounds. Its broad compatibility with industrial processes demonstrates its versatility and reliability as a raw material.
Properties That Influence Safety and Handling
Benzoic acid is generally safe to handle in controlled environments, but like many chemical compounds, it requires thoughtful storage and use. Its crystalline powder can cause mild irritation if inhaled or handled carelessly, and its acidic nature may affect sensitive tissues. In industrial applications, workers use proper protective equipment to avoid prolonged exposure. The compound’s stability ensures that it remains effective and safe when used in food preservation at regulated levels. Understanding its interactions with heat, solvents, and other chemicals helps ensure that benzoic acid remains a dependable material wherever it is applied.
Why Benzoic Acid Remains a Cornerstone Aromatic Compound
Benzoic acid’s wide-ranging importance comes from its blend of aromatic stability, acidic reactivity, and practical functionality. It bridges natural and synthetic worlds, appearing in plant-derived substances while also serving as a crucial ingredient in manufactured goods. Its structure offers chemists a reliable platform for building more complex molecules, while its antimicrobial properties support food safety and preservation. Through its role in industry, biology, and chemistry, benzoic acid demonstrates how elegantly simple organic molecules can form the foundation for technologies, processes, and natural systems that shape modern life.