Bicarbonate — Chemical Ion Structure
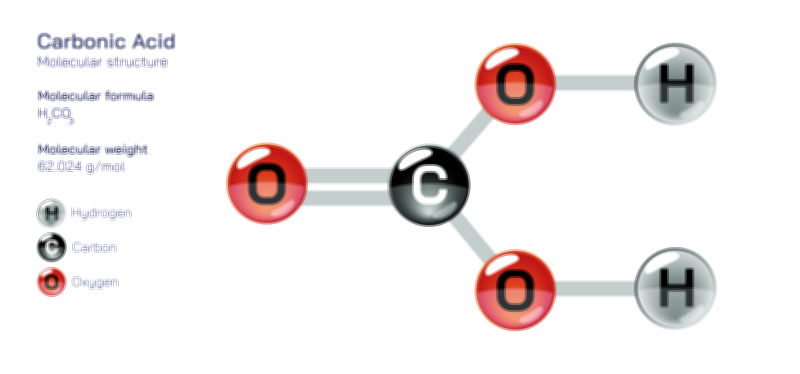
Bicarbonate is one of the most important ions found throughout natural systems, biological processes, and industrial applications, and its relevance comes from the way it balances between different chemical forms while maintaining stability within a wide range of environments. When describing bicarbonate in an explanatory manner without mathematical expressions, it is helpful to focus on how this ion behaves, where it appears, and why its structural nature allows it to play so many critical roles. At its core, bicarbonate is derived from carbonic acid and exists as a central component of the carbon cycle, making it an essential contributor to buffering processes and environmental equilibrium. When carbon dioxide dissolves in water, it undergoes a transformation that eventually produces bicarbonate, which becomes the dominant form under many natural conditions. This structural adaptability allows it to interact easily with minerals, metals, biological molecules, and atmospheric components, giving it a central place in the chemistry of life and the chemistry of the planet.
The ion itself is recognized for being highly responsive to the acidity or alkalinity of its surroundings. In more acidic conditions, bicarbonate tends to convert toward carbonic acid, while in more alkaline environments it moves toward carbonate, demonstrating its ability to shift forms depending on the needs of the environment. This characteristic makes bicarbonate a crucial part of natural buffering systems such as those found in rivers, oceans, soils, and even within the human body. Inside living organisms, bicarbonate helps maintain stable internal conditions, especially in blood where delicate balance is essential for proper functioning. The body relies on the bicarbonate buffering system to prevent harmful swings in acidity, allowing enzymes, cells, and biochemical pathways to operate within narrow limits. Its ease of formation and transformation allows the body to use it as a rapid-response regulator, constantly adjusting to changes caused by respiration, metabolism, and external factors.
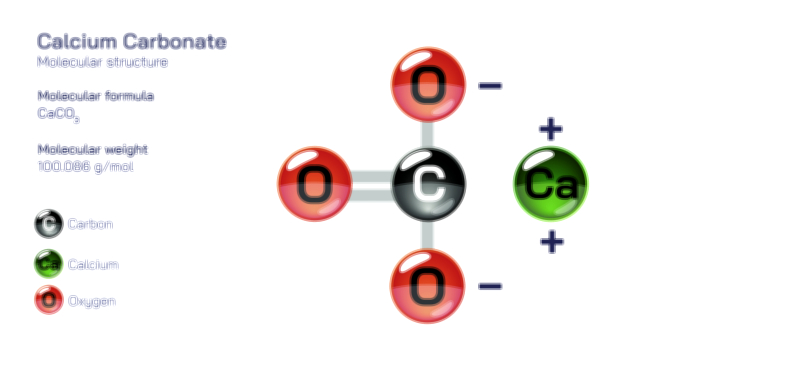
Beyond biological systems, bicarbonate plays a major role in environmental chemistry. Natural waters contain varying amounts of bicarbonate depending on the geological formations they flow through. Limestone regions tend to enrich water with bicarbonate because of the natural interaction between water, carbon dioxide, and calcium-containing minerals. This process contributes to the formation of caves, sinkholes, and other geological structures as bicarbonate supports mineral dissolution and reprecipitation over long periods. In ocean chemistry, bicarbonate is essential for maintaining the balance of dissolved inorganic carbon, which influences marine life, coral formation, and global climate cycles. The ocean acts as the largest reservoir of bicarbonate on Earth, allowing it to buffer atmospheric carbon dioxide variations and slow the pace of changes in climate systems.
In industrial and practical contexts, bicarbonate is familiar in products like baking soda, fire extinguishers, detergents, and medicinal antacids. These uses stem from its structural stability and mild reactivity, which allow it to neutralize acids, release gases under specific conditions, or gently adjust pH without causing harsh reactions. Because it is relatively safe, environmentally friendly, and widely available, bicarbonate is incorporated into many daily-use products where controlled chemical behavior is needed. Its ability to interact predictably with moisture, heat, and mild acids makes it a versatile compound in both household and commercial formulations.
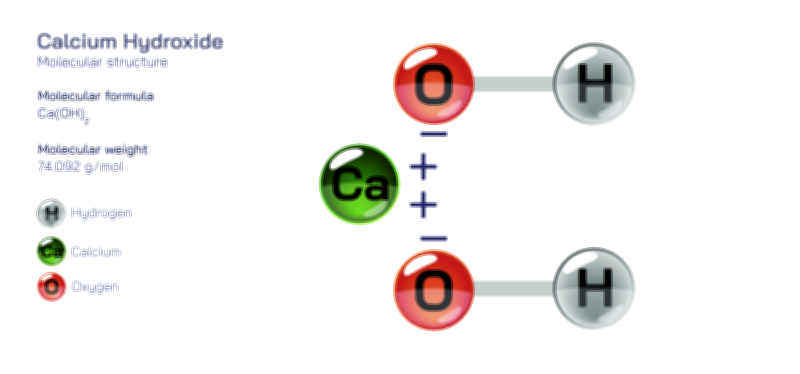
Structurally, bicarbonate is best understood as an ion with a central carbon atom bonded to oxygen atoms in a stable arrangement that supports its ability to shift between different forms without needing extreme conditions. While avoiding mathematical or symbolic descriptions, one can say that the carbon sits in the center of a triangular pattern of oxygen atoms, with one of those oxygen atoms carrying a negative charge that gives the bicarbonate ion its overall behavior. This arrangement allows the ion to share and distribute its internal forces in a way that stabilizes it even as it participates in chemical exchanges. The strength and flexibility of this structure are what make bicarbonate such a reliable participant in environmental and biological equilibrium systems. It holds enough stability to persist in solution, yet it remains reactive enough to contribute to processes like neutralization, buffering, and mineral transformation.
In everyday life, people encounter bicarbonate without realizing how central its structural nature is to the world around them. From the air they exhale, which contains carbon dioxide that will soon convert to bicarbonate in water, to the stability of their blood, which depends on bicarbonate to prevent harmful shifts in acidity, the ion sustains balance in countless ways. It regulates ecosystems, shapes geological landscapes, supports biological survival, enhances industrial processes, and acts as one of the most widely distributed and adaptable chemical forms derived from carbon. Its structure gives it the unique capability to bridge the worlds of solid minerals, dissolved gases, and living tissues, making bicarbonate a quiet but powerful component of nearly every natural and human-influenced system.