Ammonium Bromide — Structure, Properties, and Chemical Behavior
Understanding the Nature of Ammonium Bromide
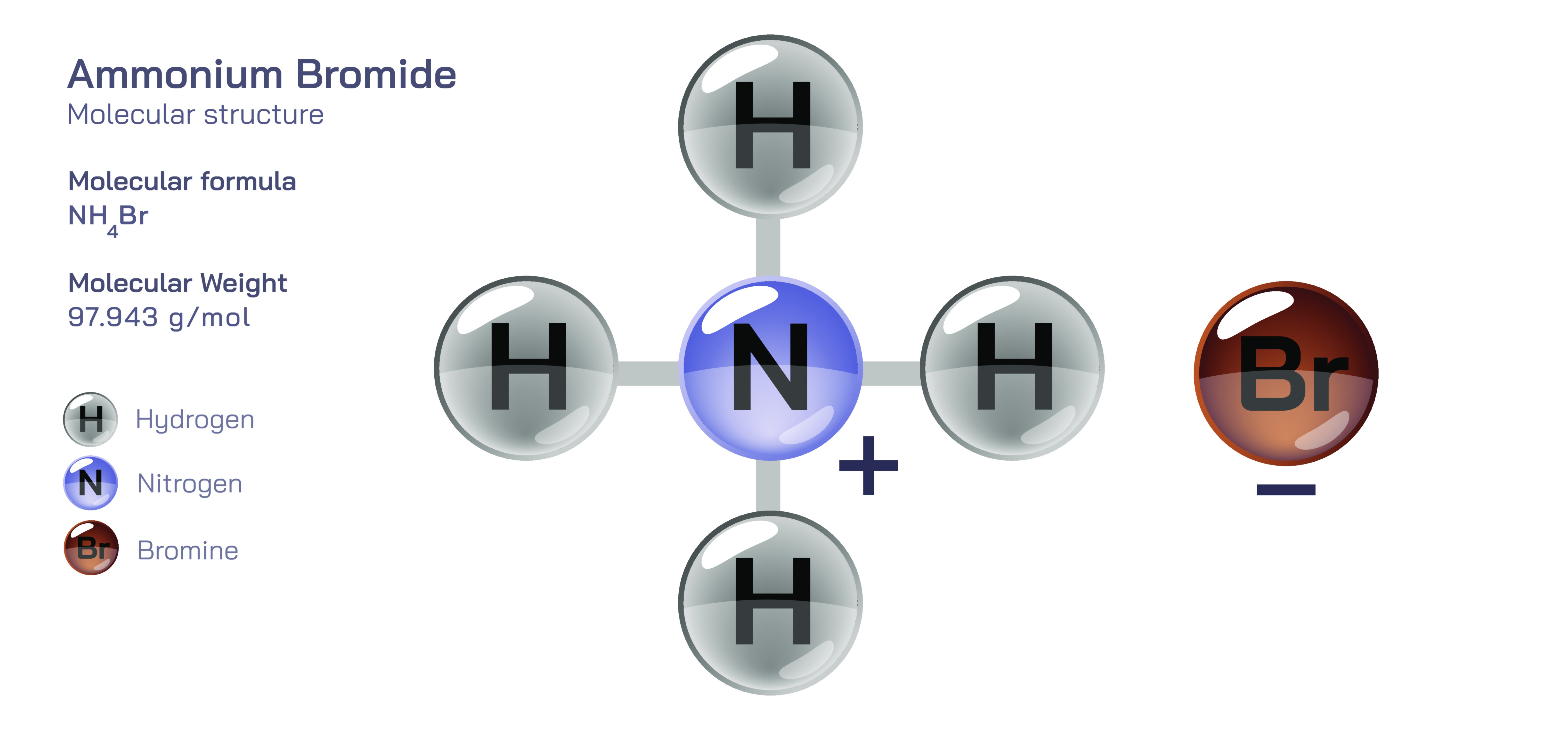
Ammonium bromide is a simple yet versatile inorganic salt formed through the pairing of ammonium ions and bromide ions. It appears as a white crystalline solid with a smooth, slightly glossy texture and dissolves easily in water to form a clear solution. Like other ammonium halides, it carries the recognizable sharp scent of ammonia when heated or exposed to moist air due to gradual decomposition. Despite its uncomplicated structure, ammonium bromide plays an important role in photography, pharmaceuticals, chemical synthesis, and laboratory testing. It is valued for its predictable behavior, high solubility, and the balanced combination of the ammonium group's mild reactivity with the bromide ion’s stable, halogen-based characteristics. In vector illustrations, the compound is often depicted by pairing the compact, symmetric ammonium group with the larger, singular bromide ion, giving viewers a clear visual sense of its ionic identity and structural simplicity.
The Ammonium Component and Its Contribution to the Salt
At the core of ammonium bromide lies the ammonium ion, an essential building block formed when ammonia binds an extra hydrogen. This creates a stable, symmetrical arrangement of one nitrogen and four hydrogens, resulting in a positively charged group that behaves like a typical cation in many inorganic salts. In ammonium bromide, this ammonium unit influences the compound’s solubility, reactivity, and mild acidity when dissolved in water. It releases a small amount of ammonia in moist conditions, giving the salt a characteristic odor. In visual representations, the ammonium group is shown as a compact central cluster with evenly spaced hydrogens, conveying both its shape and its ability to pair cleanly with bromide. This structure is responsible for many of the salt’s reactive features, especially in conditions involving heat or moisture.
The Role of the Bromide Ion and Its Chemical Stability
Bromide is a stable halide ion known for its ability to participate in substitution reactions while remaining relatively gentle and non-reactive compared to its more aggressive halogen relatives. In ammonium bromide, the bromide ion serves as the balancing partner to the ammonium ion, forming a solid lattice held together by ionic attraction. Its presence contributes to the salt’s high solubility and allows ammonium bromide solutions to interact with metals, organic molecules, and oxidizing agents in predictable, controlled ways. Over time, bromide remains chemically unchanged, even as the ammonium portion may slowly release ammonia under warm or humid conditions. In vector diagrams, the bromide ion is often shown as a single large sphere or shaded element, helping illustrate its identity as a stable, monatomic anion within the crystal structure.
Physical Appearance, Solubility, and Thermal Behavior
Ammonium bromide typically appears as smooth white crystals or granules that dissolve rapidly in water, producing solutions used for printing processes, chemical reactions, and medical formulations. The crystals are stable under normal storage conditions, but when heated or exposed to strong moisture, the compound begins to decompose, releasing ammonia and hydrogen bromide vapors. This decomposition is gradual at first but accelerates with heat, which is why the salt is stored in sealed containers and kept away from prolonged exposure to humidity. The salt has no distinct taste when fresh, but its decomposition products give it a pungent, ammoniacal scent. In vector illustrations, these properties may be symbolized through sharp crystal shapes, dissolving particles, or gentle vapor hints, offering a conceptual portrayal of its physical and reactive nature.
Chemical Behavior and Reaction Tendencies
In solution, ammonium bromide produces an environment that is mildly acidic due to the presence of ammonium ions. This makes the compound useful in applications where controlled acidity or bromide availability is needed. It can react with strong bases to release ammonia, participate in organic synthesis reactions that require bromide as a leaving group, or combine with metal ions to create specific bromide-containing products. Its predictable dissociation in water gives it a reliable role in analytical chemistry, where the presence of bromide ions is essential for certain tests or precipitation reactions. The salt also interacts with oxidizing agents to form bromine compounds, a transformation used in research and industrial processes where bromine chemistry is required.
Applications in Photography, Pharmaceuticals, and Industry
Ammonium bromide has a long history in photographic development, where bromide ions help regulate the light-sensitive reactions that form images on photographic film. It also appears in certain sedative formulations, though these uses are far less common today due to the development of modern pharmaceuticals. In the chemical industry, ammonium bromide serves as a reagent in dye-making, corrosion testing, flame retardant formulations, and specialized synthesis work that relies on bromide availability. Its high solubility and consistent behavior make it an ideal choice for laboratory studies that require a dependable source of bromide without introducing heavy metals or complex anionic structures.
Vector Illustration Representation of Ammonium Bromide
In vector illustrations, ammonium bromide is often shown as a pairing of two distinct components: the ammonium group, typically drawn as a compact geometric cluster, and the bromide ion, shown as a larger, singular sphere. This visual contrast helps communicate the ionic nature of the compound and the roles each component plays. Some diagrams also portray the crystalline lattice as repeating patterns or show the salt dissolving into separate ions in water. These visuals help simplify the compound’s chemistry while making its structure and behavior more intuitive for learners.