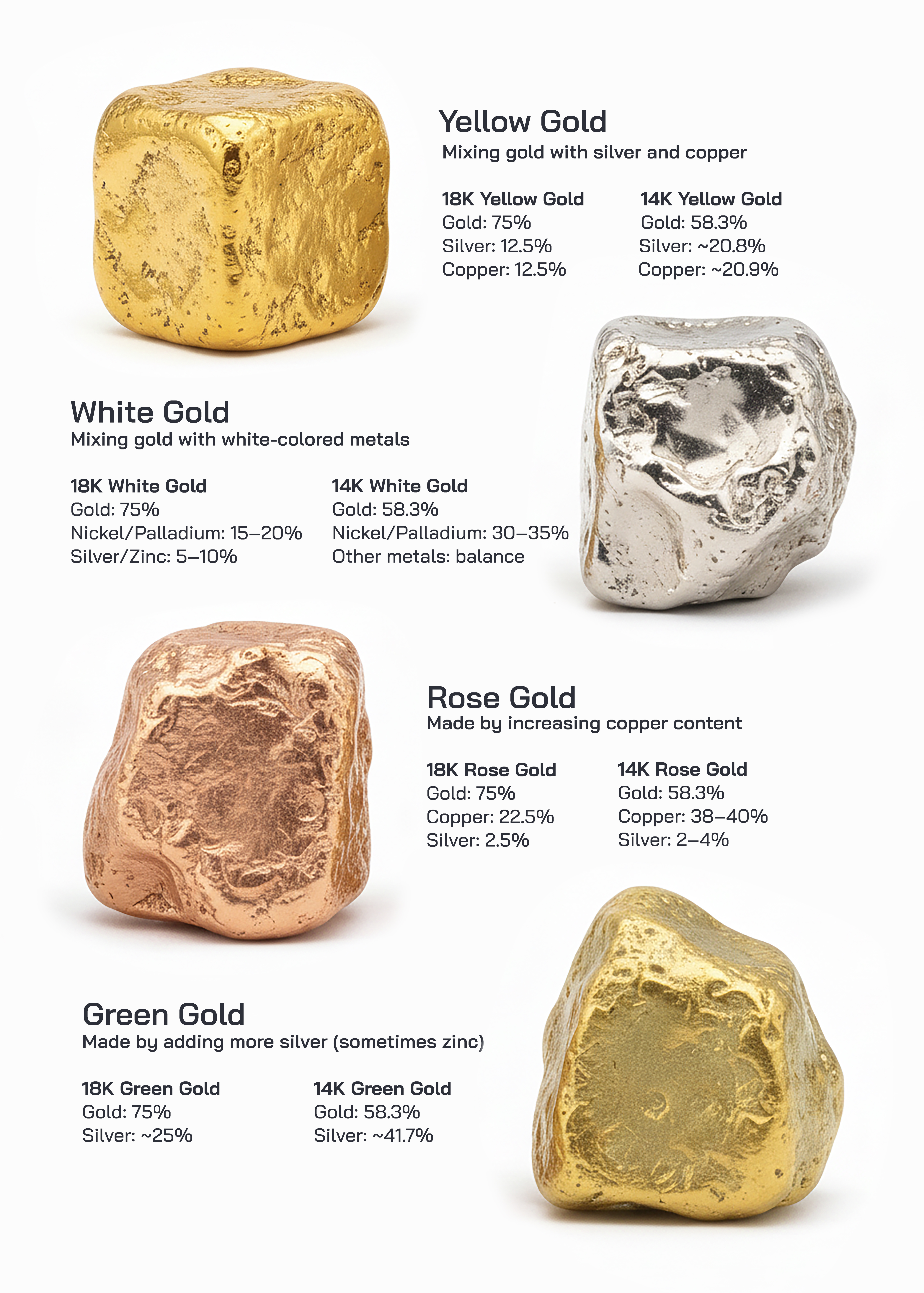
Gold has been valued by human civilizations for thousands of years due to its beauty, rarity, and resistance to corrosion. In its pure form, gold is a bright yellow metal that is soft and highly malleable. Because pure gold is too soft for most practical uses, especially jewelry, it is commonly mixed with other metals to improve strength, durability, and wear resistance. These mixtures are known as gold alloys. By varying the type and proportion of alloying metals, gold can be produced in different colors, including yellow, white, rose, and green. Although these gold types differ in appearance and properties, they are all based on the same fundamental element, gold, and are classified according to their karat value.
The karat system is used to measure the purity of gold. Pure gold is defined as 24 karat, meaning it contains nearly 100 percent gold. Lower karat values indicate that gold has been mixed with other metals. For example, 18 karat gold contains 75 percent gold and 25 percent other metals, while 14 karat gold contains approximately 58.3 percent gold and 41.7 percent alloy metals. The remaining percentage consists of metals such as silver, copper, nickel, palladium, or zinc, depending on the desired color and properties.
Yellow gold is the most traditional and widely recognized form of gold. It closely resembles the natural color of pure gold and has been used in jewelry and ornaments for centuries. Yellow gold is produced by mixing gold with silver and copper in balanced proportions. In 18 karat yellow gold, the alloy typically consists of about 75 percent gold, with the remaining 25 percent divided equally between silver and copper. This combination maintains the classic yellow color while improving hardness and durability. In 14 karat yellow gold, gold content is reduced to about 58.3 percent, while silver and copper make up the remainder, resulting in a slightly paler yellow tone and increased strength. Yellow gold is valued for its traditional appearance, resistance to tarnish, and suitability for both modern and antique-style jewelry.
White gold is created by alloying gold with white-colored metals to produce a silvery appearance. Common alloying metals used in white gold include nickel, palladium, silver, and zinc. In 18 karat white gold, gold typically makes up 75 percent of the alloy, while the remaining portion includes nickel or palladium combined with small amounts of silver or zinc. In 14 karat white gold, gold content drops to approximately 58.3 percent, and the proportion of white metals increases, enhancing hardness and durability. White gold does not naturally have a bright white finish, so it is often coated with rhodium, a noble metal that enhances shine and scratch resistance. White gold is popular in contemporary jewelry, particularly for diamond settings, because its neutral color enhances gemstone brilliance.
Rose gold, also known as pink gold, is distinguished by its warm reddish hue. This color is achieved by increasing the proportion of copper in the gold alloy. In 18 karat rose gold, the alloy typically contains about 75 percent gold, approximately 22.5 percent copper, and a small amount of silver to balance the tone. In 14 karat rose gold, gold content is around 58.3 percent, while copper content can rise to nearly 38 to 40 percent, with minimal silver. The high copper content gives rose gold its distinctive color and also increases hardness, making it more durable than yellow gold. Rose gold has gained popularity in modern jewelry due to its romantic appearance, vintage appeal, and excellent wear resistance.
Green gold is a less common but historically significant gold alloy known for its subtle greenish-yellow tint. Green gold is produced by alloying gold primarily with silver and sometimes small amounts of zinc. Unlike rose or white gold, green gold does not rely on copper or nickel for its color. In 18 karat green gold, gold makes up 75 percent of the alloy, while silver accounts for roughly 25 percent. In 14 karat green gold, gold content is approximately 58.3 percent, with silver making up about 41.7 percent. The resulting color is a soft greenish-yellow that is more muted than yellow gold. Green gold is softer than rose or white gold due to the absence of strengthening metals like copper or nickel, which limits its use to decorative or low-stress jewelry designs.
The differences between yellow, white, rose, and green gold are primarily determined by alloy composition rather than gold purity. Two pieces of jewelry with the same karat value can have identical gold content but very different colors and mechanical properties depending on the metals mixed with the gold. Yellow gold emphasizes balance and tradition, white gold focuses on modern aesthetics and gemstone enhancement, rose gold highlights warmth and durability, and green gold offers a unique, subtle color variation rooted in historical metallurgy.
Karat value also affects performance and usage. Higher karat gold contains more gold and is softer, making it suitable for ornamental or ceremonial jewelry. Lower karat gold contains more alloy metals, increasing hardness and making it more practical for daily wear. For this reason, 18 karat gold is often chosen for luxury jewelry, while 14 karat gold is preferred for rings, bracelets, and items exposed to frequent handling.
Each gold type has distinct advantages in terms of maintenance and longevity. Yellow gold requires minimal surface treatment and does not rely on plating. White gold may need periodic rhodium replating to maintain its bright appearance. Rose gold retains its color naturally and resists scratching due to copper content. Green gold, while visually unique, requires careful handling because of its softer nature.
Beyond color and purity, gold alloys exhibit distinct mechanical and metallurgical behaviors that influence how they are used in jewelry, technology, and industry. Gold’s atomic structure allows it to bond easily with many metals, and these bonds alter not only color but also hardness, elasticity, melting temperature, and resistance to deformation. The selection of alloying elements is therefore a balance between aesthetics and functional performance, shaped by centuries of experimentation and modern material science.
Historically, different gold colors emerged in response to regional metallurgy and available resources. Ancient civilizations did not categorize gold by color in the modern sense, but variations naturally occurred depending on local copper and silver content. Greenish and pale yellow gold alloys were common in ancient Mediterranean cultures where silver-rich ores were abundant. Rose-toned gold appeared more frequently in regions with higher copper availability, long before it became a stylistic choice. White gold is comparatively modern, emerging in the twentieth century as a response to demand for platinum-like appearance at lower cost.
From a metallurgical standpoint, alloying gold alters its crystal lattice. Pure gold has a face-centered cubic structure that allows extreme malleability. Introducing copper, silver, or nickel distorts this lattice, restricting atomic movement and increasing strength. Copper atoms, being slightly smaller, create internal stress that hardens the alloy, while silver atoms preserve softness but modify reflectivity. These microscopic structural changes explain why rose gold resists bending better than green gold and why white gold requires surface treatment for brightness.
Thermal behavior also differs among gold alloys. Melting temperatures vary depending on composition, which affects casting and manufacturing techniques. Alloys with higher copper content melt at lower temperatures than those with higher silver or palladium content. This influences mold design, cooling rates, and grain structure during solidification. Improper cooling can lead to internal stresses or brittleness, making alloy control critical in precision jewelry and industrial components.
Cultural symbolism plays a major role in the preference for different gold colors. Yellow gold has long symbolized wealth, power, and permanence due to its association with the sun and immortality in ancient cultures. White gold conveys modernity, purity, and technological refinement, often linked to contemporary design and innovation. Rose gold has been associated with romance, warmth, and artistic expression, while green gold historically symbolized fertility, nature, and renewal, reflecting its subtle organic hue.
Skin interaction is another practical consideration. Certain alloying metals can cause allergic reactions in sensitive individuals. Nickel-based white gold alloys are known to cause skin irritation in some wearers, leading to increased use of palladium-based alternatives. Yellow and rose gold are generally better tolerated, while green gold’s high silver content can sometimes cause mild discoloration on skin under humid conditions. These factors influence alloy selection for wearable items.
Gold alloys also differ in long-term wear characteristics. Copper-rich alloys develop a natural patina over time, subtly deepening rose gold’s color. White gold relies on surface coatings to maintain appearance, making it more susceptible to visible wear patterns. Yellow gold shows scratches more easily due to its reflective surface but is simpler to polish and restore. Green gold, being softer, may deform under stress, limiting its use in structural jewelry components.
In modern industry, colored gold alloys extend far beyond ornamentation. Gold alloys are used in electronics for corrosion-resistant connectors, microcircuits, and bonding wires. Alloy composition is carefully controlled to balance conductivity, hardness, and thermal expansion. Even slight variations in alloying elements can affect electrical resistance and mechanical reliability in precision applications.
Recycling and sustainability are increasingly important aspects of gold alloy use. Gold can be recycled indefinitely without loss of quality, and alloyed gold is routinely refined back into pure form before re-alloying. This process reduces environmental impact and allows the same gold atoms to be reused across centuries in different forms. Colored gold alloys therefore represent not just aesthetic variation, but adaptable material states within a closed-loop resource system.
Modern computational metallurgy has further advanced gold alloy development. Using predictive modeling, scientists can simulate how different alloy combinations affect color perception, strength, and durability before physical production. This allows creation of new shades and performance profiles tailored to specific applications, blending ancient metalworking traditions with cutting-edge technology.
Gold alloys exhibit distinct optical behaviors that determine how color is perceived by the human eye. The color of gold is not merely a surface effect but arises from interactions between light and the electronic structure of the alloy. Pure gold absorbs blue light and reflects yellow and red wavelengths, giving it its characteristic color. When alloying metals are introduced, they alter electron energy levels, subtly shifting which wavelengths are absorbed or reflected. Silver increases reflectivity across the visible spectrum, lightening the appearance, while copper increases absorption in the green-blue range, intensifying red and pink hues. Green gold emerges when silver dominates without copper, preserving brightness while slightly shifting hue.
The surface microstructure of gold alloys also influences visual appearance. Polishing, grain size, and surface orientation affect how light scatters. Rose gold tends to diffuse light more softly due to copper-rich grain boundaries, while white gold reflects light more sharply after rhodium plating. Green gold produces a satin-like luster rather than a mirror finish, which contributes to its understated aesthetic. These differences explain why identical karat values can appear dramatically different under the same lighting conditions.
Mechanical aging is another important aspect of gold alloys. Over time, repeated stress causes microscopic deformation known as work hardening. Alloys containing copper harden more rapidly, increasing scratch resistance but reducing flexibility. This is why rose gold jewelry often becomes more rigid with long-term wear. Yellow gold retains flexibility longer, making it easier to resize or repair even after decades. Green gold, being silver-rich, is more susceptible to fatigue and microcracking if subjected to repeated bending, which limits its structural applications.
Historically, alloy selection was influenced by available smelting technology. Early goldworkers lacked precise control over composition, resulting in naturally alloyed gold with variable colors. Ancient texts describe “electrum,” a naturally occurring gold–silver alloy that predates modern green gold. Electrum was widely used in early coinage because it was harder than pure gold and easier to mint. These historical alloys laid the groundwork for deliberate color engineering in later eras.
From a thermodynamic perspective, gold alloys behave differently under heat exposure. Copper-rich alloys oxidize more readily at high temperatures, forming surface oxides that must be removed during manufacturing. Silver-rich alloys resist oxidation but can suffer from grain growth if overheated, reducing mechanical strength. White gold alloys containing palladium exhibit higher melting stability, making them suitable for precision casting. These thermal behaviors dictate manufacturing workflows in jewelry and industrial fabrication.
Gold color also plays a psychological and cultural role in consumer preference. Studies in material perception show that warmer gold tones are associated with emotional warmth and tradition, while cooler tones are associated with modernity and precision. This perception influences design trends in jewelry, architecture, and luxury goods. Designers often select gold color to complement skin tone, lighting environment, and intended emotional impact rather than solely for durability.
Investment and valuation considerations further differentiate gold alloys. While all karat gold contains intrinsic gold value, market pricing may vary due to alloy desirability, manufacturing cost, and maintenance requirements. White gold pieces may incur additional long-term costs due to replating, while rose gold’s durability can increase resale appeal. Green gold, though rare, may command niche interest due to historical or artisanal significance rather than metal value alone.
In contemporary design science, gold alloys are increasingly evaluated using sustainability metrics. Copper and silver sourcing has different environmental footprints, influencing alloy choice in ethically driven manufacturing. Palladium-based white gold, while hypoallergenic, involves rarer metals with higher extraction impact. These considerations are shaping new alloy research aimed at balancing aesthetics, performance, and environmental responsibility.
Technological innovation continues to expand the boundaries of gold alloy usage. Nanostructured gold alloys are being explored for biomedical implants, sensors, and advanced electronics. At nanoscale dimensions, color behavior changes dramatically due to plasmonic effects, allowing gold alloys to appear red, green, or even blue without traditional alloying. These developments demonstrate that gold’s color versatility extends beyond classical metallurgy into advanced physics.
Across history and modern science, gold alloys represent a dialogue between nature and human ingenuity. Each color variant is not merely decorative but embodies deliberate manipulation of atomic structure, light interaction, mechanical behavior, and cultural meaning. The enduring appeal of yellow, white, rose, and green gold lies in their ability to adapt to changing human needs while preserving the elemental permanence that has defined gold’s value for millennia.
Ultimately, yellow, white, rose, and green gold are expressions of gold’s remarkable adaptability rather than separate materials. Each alloy represents a deliberate modification of gold’s intrinsic properties to meet cultural preferences, functional requirements, and technological needs. The enduring value of gold lies not only in its purity but in its ability to transform while retaining its fundamental identity, making it one of the most versatile and enduring materials known to humanity.
In summary, yellow, white, rose, and green gold are all gold alloys differentiated by the metals mixed with gold and their resulting color, strength, and appearance. The karat system defines gold purity, while alloy composition determines physical and aesthetic properties. Yellow gold combines gold with silver and copper for a classic look, white gold uses white metals for a modern finish, rose gold relies on copper for warmth and durability, and green gold uses silver to create a subtle greenish tone. Understanding these differences helps in selecting the appropriate gold type for jewelry, investment, and decorative purposes, highlighting the versatility and enduring value of gold as a precious metal.