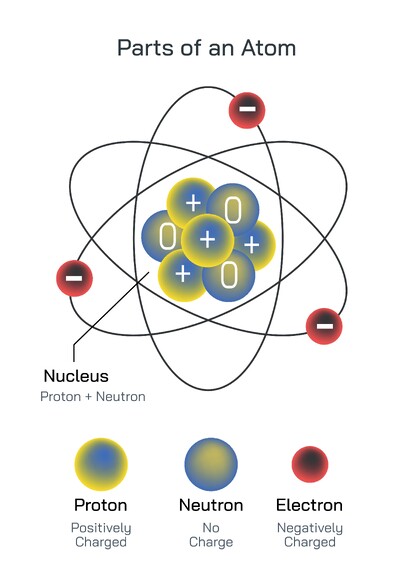
Sublevels and atomic orbitals are central ideas in modern atomic theory and quantum chemistry. They explain how electrons are arranged around the nucleus of an atom and why atoms behave the way they do during chemical reactions. In the quantum mechanical model of the atom, electrons are not treated as particles moving in fixed circular paths. Instead, they are described by wave functions that define regions of space where electrons are most likely to be found. These regions are called atomic orbitals. Among all orbitals, the most fundamental and widely studied are the s and p orbitals, which together explain a large part of atomic structure, periodic trends, and chemical bonding.
An atomic sublevel, also known as a subshell, is a subdivision of an energy level. Each sublevel contains one or more atomic orbitals that have similar energy and shape. The s and p sublevels are the first and second types of sublevels encountered as electrons occupy an atom. Understanding these two orbitals provides a strong foundation for learning atomic structure and chemistry.
The s orbital is the simplest and lowest-energy type of atomic orbital. It is present in every principal energy level of an atom. The most distinctive feature of an s orbital is its spherical shape. In an s orbital, the probability of finding an electron depends only on the distance from the nucleus and not on the direction. This means that the electron cloud is evenly distributed in all directions around the nucleus, forming a symmetrical sphere.
Because of this spherical symmetry, s orbitals have no directional orientation along the x, y, or z axes. Whether an electron is found above, below, or to the side of the nucleus, the probability remains the same at a given distance. This property makes s orbitals especially important in understanding the core structure of atoms and the formation of sigma bonds in chemistry.
The s sublevel contains only one orbital, and each orbital can hold a maximum of two electrons. These electrons must have opposite spins, in accordance with the Pauli exclusion principle. As the principal energy level increases, the size of the s orbital also increases. Higher-energy s orbitals are larger and more diffuse, meaning the electron cloud extends farther from the nucleus.
Although all s orbitals are spherical, their electron density distribution changes with energy level. In higher s orbitals, regions of higher and lower electron probability appear at different distances from the nucleus. These variations do not change the overall spherical shape but reflect the wave nature of electrons in quantum mechanics.
The p orbitals represent the next level of complexity in atomic structure. The p sublevel appears from the second energy level onward. Unlike the s sublevel, the p sublevel contains three distinct orbitals. These orbitals are oriented along three mutually perpendicular axes and are known as px, py, and pz orbitals.
The most characteristic feature of p orbitals is their dumbbell shape. Each p orbital consists of two lobes on opposite sides of the nucleus, separated by a region of very low electron probability at the nucleus itself. This region is known as a nodal plane. The presence of a node is a key difference between s and p orbitals and reflects a change in the sign of the wave function.
Each p orbital has a specific orientation in space. The px orbital is aligned along the x-axis, the py orbital along the y-axis, and the pz orbital along the z-axis. Although they differ in orientation, all three p orbitals within the same sublevel have the same energy in an isolated atom. This equality in energy is known as degeneracy.
The directional nature of p orbitals plays a crucial role in chemical bonding. Because p orbitals point in specific directions, they allow atoms to form directional bonds. This property explains the shapes of molecules and the angles between bonds. For example, the orientation of p orbitals helps determine whether a molecule is linear, trigonal, or tetrahedral in structure.
Like s orbitals, each p orbital can hold a maximum of two electrons with opposite spins. Since there are three p orbitals in a p sublevel, the p sublevel can accommodate a total of six electrons. The distribution of electrons among these orbitals follows Hund’s rule, which states that electrons occupy orbitals singly with parallel spins before pairing occurs. This rule minimizes electron repulsion and contributes to atomic stability.
The energy of p orbitals is generally higher than that of s orbitals within the same energy level. This difference in energy influences the order in which electrons fill orbitals as atomic number increases. The relative energies of s and p orbitals also affect ionization energy, atomic size, and reactivity trends across the periodic table.
From a probability perspective, atomic orbitals do not represent fixed paths but regions where the likelihood of finding an electron is highest. The shapes of s and p orbitals are visual representations of these probability distributions. In an s orbital, the probability density is highest near the nucleus and decreases smoothly with distance. In p orbitals, the probability density is concentrated in two distinct regions on opposite sides of the nucleus.
The quantum mechanical description of s and p orbitals provides a more accurate picture of atomic structure than earlier models. It explains why atoms emit and absorb specific energies, why elements form characteristic chemical bonds, and why certain electron configurations are more stable than others. The wave nature of electrons, combined with the shapes and orientations of orbitals, forms the foundation of modern chemistry and physics.
In chemical bonding, s and p orbitals combine to form hybrid orbitals, which further explain molecular geometry. However, even before introducing hybridization, understanding pure s and p orbitals is essential. They describe how electrons are arranged in isolated atoms and how valence electrons participate in bonding.
In educational contexts, diagrams showing s and p orbitals are widely used to help students visualize atomic structure. These diagrams illustrate the spherical symmetry of s orbitals and the directional dumbbell shapes of p orbitals, making abstract quantum concepts more tangible. Such visual tools bridge the gap between mathematical descriptions and physical understanding.
In conclusion, s and p orbitals are fundamental components of atomic sublevels and play a critical role in determining atomic behavior. The s orbital is spherical, non-directional, and capable of holding two electrons, while the p sublevel consists of three directional orbitals aligned along the x, y, and z axes, collectively holding up to six electrons. Together, s and p orbitals explain electron distribution, atomic stability, and the directional nature of chemical bonding. A clear understanding of these orbitals forms the basis for advanced studies in chemistry, physics, and material science, highlighting the quantum nature of matter and the structured organization of electrons within atoms.