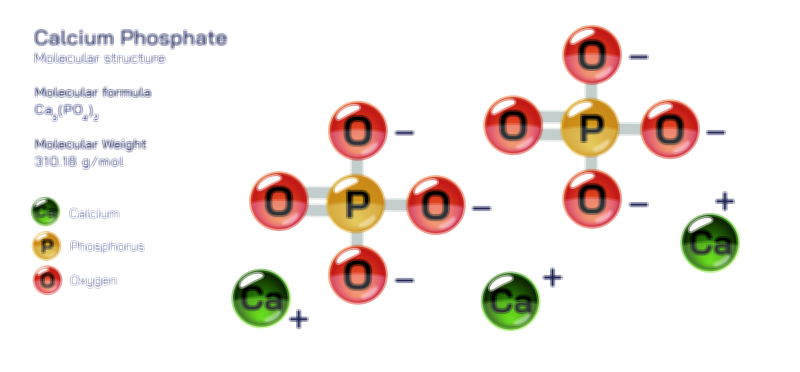
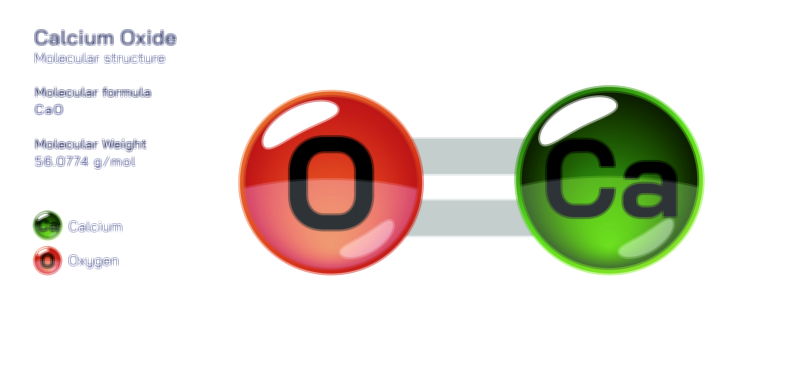
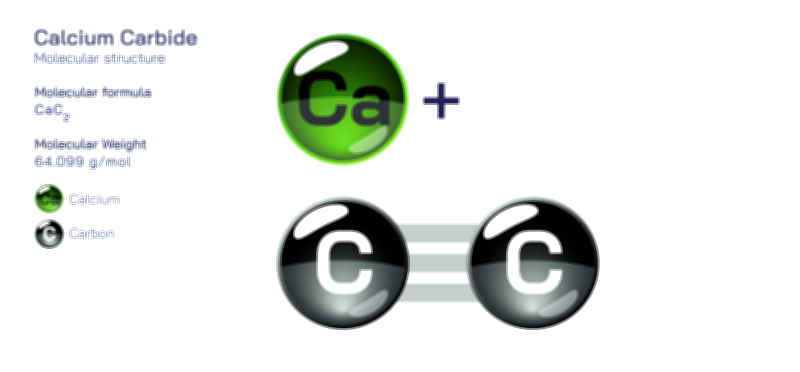
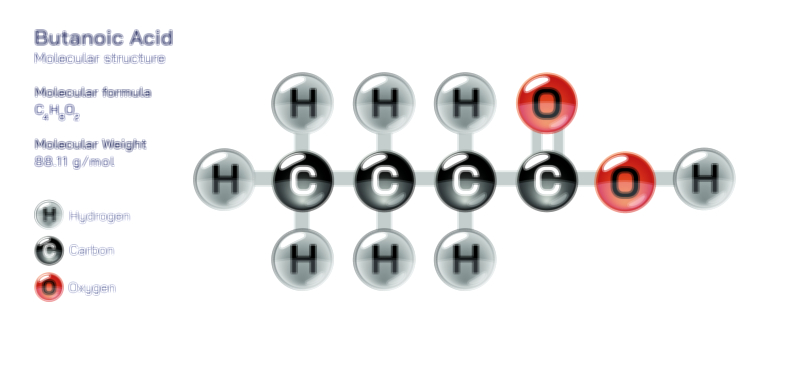
Bromine — Halogen Element Molecular Structure
Bromine occupies a unique and fascinating place among the halogens, representing a transitional point between the lighter and heavier members of its group and revealing complex chemical behavior that reflects its intermediate physical and electronic properties. As one of the only elements that exists naturally as a liquid at room temperature, bromine immediately stands apart, displaying a deep reddish-brown coloration and releasing vapors that appear intensely tinted, almost as though the substance carries its own visual identity. This distinctive appearance hints at the underlying molecular structure that contributes to bromine’s reactivity, its bond characteristics, and its ability to participate in a wide variety of chemical transformations. While molecular structures can be described visually in vector illustrations, the conceptual understanding behind those shapes lies in the way bromine atoms pair to form diatomic molecules. Each molecule consists of two bromine atoms bonded through shared electron pairs, creating a stable, symmetrical structure that reflects the halogens’ tendency to exist naturally as two-atom units. This diatomic arrangement is essential for understanding how bromine behaves in reactions, how it interacts with other elements, and how its presence influences environmental and industrial systems.
At the atomic level, bromine’s electronic configuration reveals why it is both reactive and yet more controlled compared to lighter halogens such as chlorine and fluorine. Its outer valence shell contains seven electrons, placing it just one electron short of a full, stable shell. This near-completion allows bromine to readily participate in electron-gaining reactions, forming negative ions that contribute to ionic compounds or engaging in covalent bonding when interacting with other non-metals. The diatomic structure, which is typically shown in vector illustrations as two spheres of equal size connected by a clear bond, visually captures the simplicity and symmetry of this molecular form. However, beneath this simplicity lies the nuanced behavior of bromine’s electrons, which are more diffuse and more polarizable than those of lighter halogens. This increased polarizability contributes to bromine’s physical state as a liquid under standard conditions, since the temporary attractions between bromine molecules are strong enough to overcome the complete dispersal seen in gases but still weaker than the rigid forces found in solids.
The liquid state of elemental bromine gives rise to many of its practical implications, especially in terms of handling, environmental impact, and industrial use. Its tendency to evaporate readily means that bromine must be managed carefully, as the vapors can be irritating or harmful at sufficient concentrations. Yet this volatility also enables its usefulness in chemical synthesis, where bromine can diffuse rapidly into reaction systems. In an industrial context, bromine serves as a key ingredient in manufacturing flame retardants, pharmaceuticals, dyes, agricultural chemicals, and water-treatment agents. In many of these applications, the diatomic bromine molecule acts as a reactive starting point that participates in controlled halogenation reactions. These processes take advantage of bromine’s ability to add across double bonds in organic compounds, substitute for hydrogen atoms in certain molecular frameworks, or undergo reduction to form bromide salts, which are far more stable and commonly used in everyday applications.
Environmental chemistry provides another dimension where bromine’s molecular structure plays a defining role. In the atmosphere and in natural water systems, bromine participates in complex cycles that involve interconversions among bromine radicals, bromide ions, and various oxidized forms. These cycles influence ozone chemistry, particularly in marine environments where bromine-containing compounds emitted by biological organisms can trigger reactions that temporarily deplete ozone in localized regions. The diatomic structure allows bromine molecules to dissociate into highly reactive atoms when exposed to sunlight or other energy sources, creating pathways for radical-driven transformations. Understanding these cycles helps scientists predict atmospheric behavior, model climate interactions, and assess the environmental consequences of bromine-based industrial emissions, especially in the context of halogenated organic compounds that may persist for long periods.
In the realm of material science, bromine’s molecular properties influence how it interacts with various substrates, how it bonds chemically with metals or organic materials, and how it contributes to surface modifications. For example, brominated compounds play a prominent role in the study of self-assembled monolayers and surface patterning because the carbon–bromine bond can serve as a reactive handle for further functionalization. When bromine atoms are incorporated into organic frameworks, they influence molecular geometry, reactivity, and polarity in ways that chemists can exploit for designing advanced functional materials. In all these applications, the basic diatomic nature of elemental bromine remains the conceptual reference point from which these more complex structural roles emerge.
From a biological perspective, bromine occupies an unusual but meaningful place. While not traditionally considered a major nutrient like sodium, calcium, or iron, bromine has gained research attention for its potential role in certain enzymatic processes and its presence in marine organisms that produce naturally occurring brominated compounds. Some marine algae and invertebrates synthesize brominated metabolites that serve protective or signaling functions. These compounds illustrate how living systems can harness the reactivity of halogens to create specialized biochemical molecules. At the same time, the biological implications of bromine exposure must be considered carefully, as high concentrations or prolonged exposure can disrupt cellular processes. This duality reflects the broader nature of many halogens: reactive and beneficial in controlled contexts but potentially harmful in excess.
One of the fascinating aspects of bromine lies in how its molecular form is depicted in scientific illustrations. Vector illustrations, widely used in educational and technical materials, represent bromine molecules as simple diatomic units, often shaded in the deep reddish tones that symbolize its real-world coloration. These illustrations emphasize symmetry, bond length, and relative atomic size, allowing viewers to visualize the balanced arrangement of two identical atoms. Although simplified, these depictions capture the core characteristics of the molecule: its nonpolar nature, its bond strength relative to other halogens, and its equilibrium between condensed and vapor states. Such illustrations help students and professionals alike grasp bromine’s placement within the periodic table and its relationship to other halogens through clear, accessible imagery.
Historically, the discovery and characterization of bromine highlighted the progression of chemical understanding during the early nineteenth century. Researchers observing the unusual redness and volatility of the substance recognized quickly that it did not fit neatly into categories already known at the time. Its eventual classification as a halogen solidified the group’s conceptual identity and extended the patterns seen in chlorine and iodine. This historical context reinforces the idea that bromine occupies a middle position within its family, both chemically and physically, exhibiting reactivity and mass properties that form a bridge between the extremes represented by the lighter and heavier members.
Ultimately, bromine stands as a compelling example of how molecular structure influences physical appearance, reactivity, environmental behavior, and technological value. Its diatomic form, captured in vector illustrations as a simple two-atom unit, anchors a complex web of chemical processes and applications that span industrial synthesis, environmental science, materials chemistry, and biological interactions. The rich coloration, the unusual liquid state, the readiness to vaporize, and the capacity for both beneficial and hazardous effects all stem from the same underlying molecular identity. By understanding bromine’s structure, one gains insight not only into the element itself but also into broader principles governing halogen chemistry, redox behavior, intermolecular forces, and the intricate relationships that define the material world.