Atomic Structure — Understanding the Foundation of Fundamental Physics
The Concept of the Atom as the Building Block of Matter
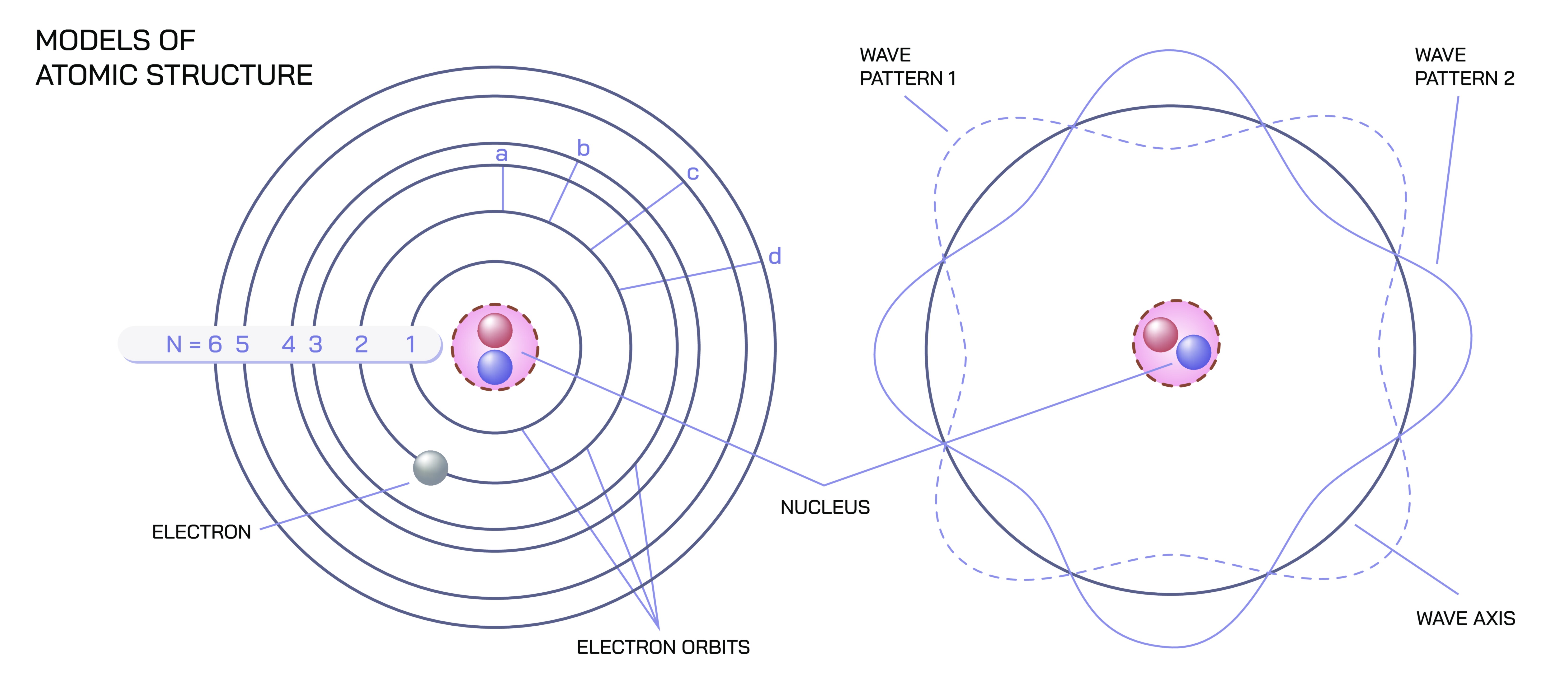
Atomic structure lies at the heart of fundamental physics, shaping our understanding of how the physical world is formed, behaves, and transforms. Every object we see, touch, or interact with is composed of atoms—tiny units so small that they cannot be observed directly with the naked eye, yet powerful enough to define the properties of all matter. The idea of the atom represents the point at which matter can no longer be divided without losing the characteristics of the substance it belongs to. Each atom consists of a central region that holds nearly all of its mass and a surrounding space that determines its interactions with other atoms. This central region, or nucleus, plays the role of an anchor, while the surrounding region shapes chemistry, bonding, and energy exchanges. Understanding this structure allows physicists and chemists to explain why materials behave differently, how particles interact, and what forces govern the stability of matter.
The Nucleus as the Dense Center of Atomic Structure
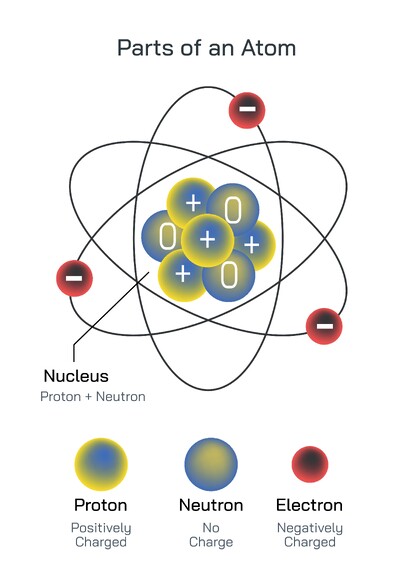
At the core of every atom is a tiny, dense nucleus composed of particles that contribute most of the atom’s weight. This nucleus is extraordinarily small compared to the full size of the atom, yet it contains the powerful internal forces that hold its components together. These forces are among the strongest in nature, binding the central particles so tightly that the nucleus remains stable under conditions that would tear apart larger structures. The density of the nucleus gives the atom its mass, while its internal arrangement influences how the atom behaves in larger systems. Even though the nucleus occupies only a small fraction of the atom’s total volume, its influence extends outward, shaping the environment in which other atomic components move. In fundamental physics, the nucleus serves as a reminder that even the smallest structures contain layers of complexity.
The Surrounding Region Where Electrons Influence Behavior
Moving outward from the nucleus brings us into the region where electrons play a vital role in the atom’s identity and interactions. These electrons exist in a cloud-like environment rather than following strict lines or pathways, forming a sort of dynamic atmosphere around the nucleus. This region is where most of an atom’s interactions with the outside world occur, especially when forming bonds with other atoms. Although electrons are extremely light compared to the nucleus, their arrangement around it determines how the atom interacts with others, whether it forms strong connections, weak associations, or remains largely unreactive. This surrounding region also gives each type of atom its signature behavior, shaping the characteristics that define familiar substances. The structure of this area is essential for understanding energy flow, the formation of molecules, and the vast range of material properties found in nature.
How Atomic Structure Shapes the Properties of Matter
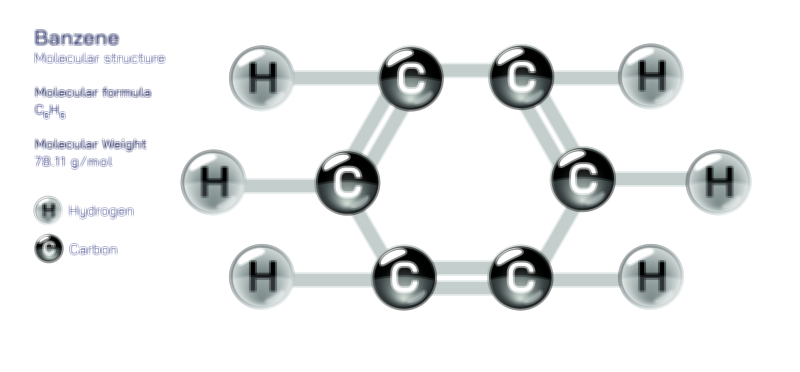
The internal arrangement of an atom directly influences the physical and chemical traits of substances. Different atoms combine in countless ways to create materials with unique behaviors—some metals shine brightly and conduct electricity, some gases remain invisible and float freely, and some solids hold their shape firmly. These differences arise because atoms interact according to how their electrons and nuclei are structured. Atoms with stable outer arrangements tend to resist change, making them less reactive, while those with incomplete arrangements eagerly combine with others. The energy levels in which electrons exist determine whether substances behave rigidly, flexibly, or in ways that allow them to conduct heat or electricity. In this sense, the atom’s structure acts as a blueprint that governs everything from the hardness of minerals to the transparency of glass and the fluidity of water.
The Role of Atomic Structure in Energy and Physical Transformations
Atomic structure also plays a crucial role in the ways matter absorbs, stores, and releases energy. By shifting their internal arrangements or moving to higher energy states, atoms can take in energy from their environment. When this energy is released, it often transforms into light or heat, creating visible and measurable physical effects. This ability for atoms to interact with energy explains why substances glow when heated, why chemical reactions occur, and why some materials can store or release energy under specific conditions. Even the colors we see in everyday life are influenced by how electrons respond to energy within the atom. In fundamental physics, these transformations help explain the relationship between matter and energy, revealing the dynamic and interconnected nature of the universe.
Atomic Structure as the Foundation of Chemical Bonding
Chemical bonding is one of the most significant outcomes of atomic structure. Atoms rarely exist alone; they come together to form molecules, crystals, and complex structures that make up the visible world. These connections occur because the electrons in the surrounding region of each atom interact in ways that allow them to share or exchange energy. Understanding atomic structure makes it possible to explain why certain atoms form strong, stable bonds while others form weaker or temporary connections. These bonding interactions allow simple atoms to generate complex materials such as proteins, minerals, fuels, and countless synthetic products. The variety of structures formed from atomic bonding demonstrates the immense potential stored within the basic building blocks of matter.
Why Atomic Structure Is Central to Fundamental Physics
Atomic structure represents the bridge between the smallest components of the universe and the large-scale behaviors we observe daily. It provides the framework for understanding how matter forms, how energy flows, and how physical changes occur. Without a clear understanding of atomic structure, it would be impossible to explain the behavior of solids, liquids, gases, or the countless materials that support life and technology. The atom’s design is simple in appearance yet filled with layers of complexity that continue to inspire scientific exploration. It sits at the foundation of physics, chemistry, materials science, astronomy, and countless other fields, showing how universal principles begin with structures so small they cannot be seen directly. Appreciating atomic structure deepens our understanding of the world’s complexity and the remarkable systems that shape the universe.