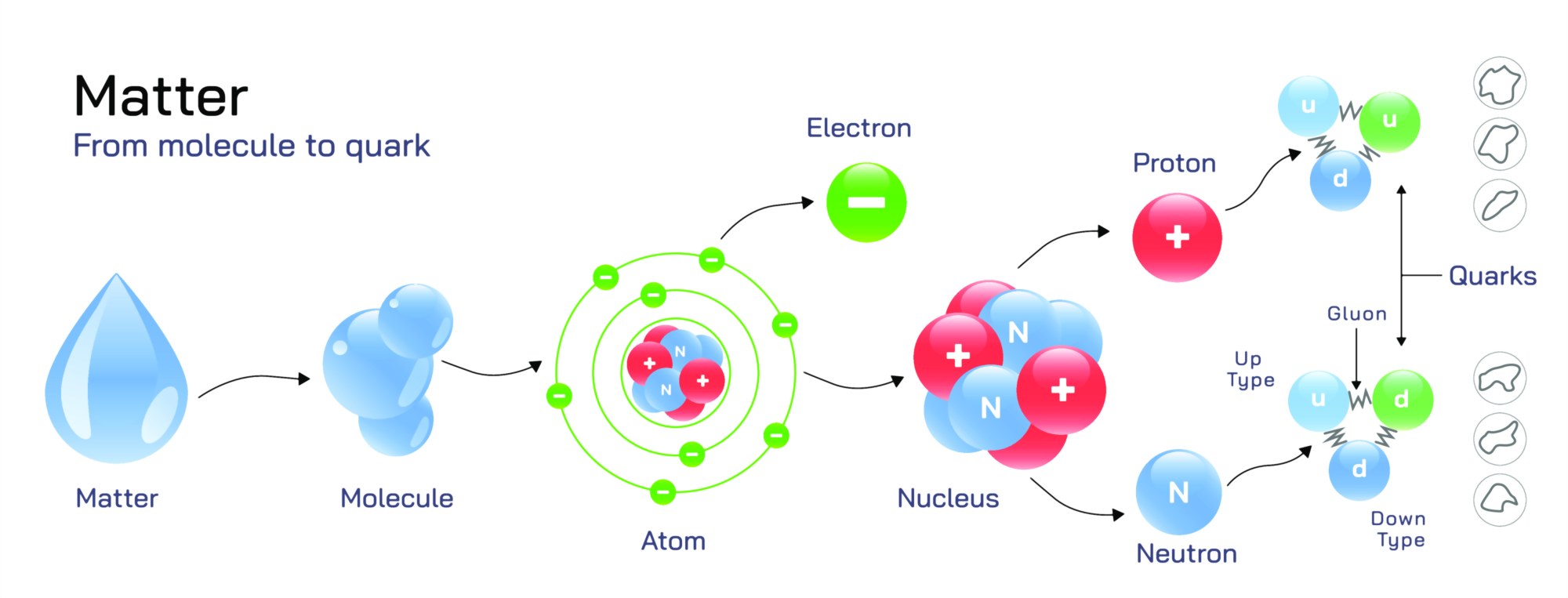
Molecule to Quark of a Water Drop Illustration: Scale from Macroscopic to Subatomic Level Explained
A drop of water is something familiar and visible to the naked eye, yet within that tiny droplet lies an astonishing hierarchy of structures ranging from molecular arrangements to the deepest subatomic constituents of matter. An illustration that zooms step-by-step from a full water drop down to the level of quarks offers a dramatic visualization of how physical reality is built through nested layers of scale—each governed by its own scientific rules and forms of organization. By moving from the macroscopic world into the atomic and then subatomic domains, such a diagram bridges chemistry, physics, quantum mechanics, and particle theory into one unified story.
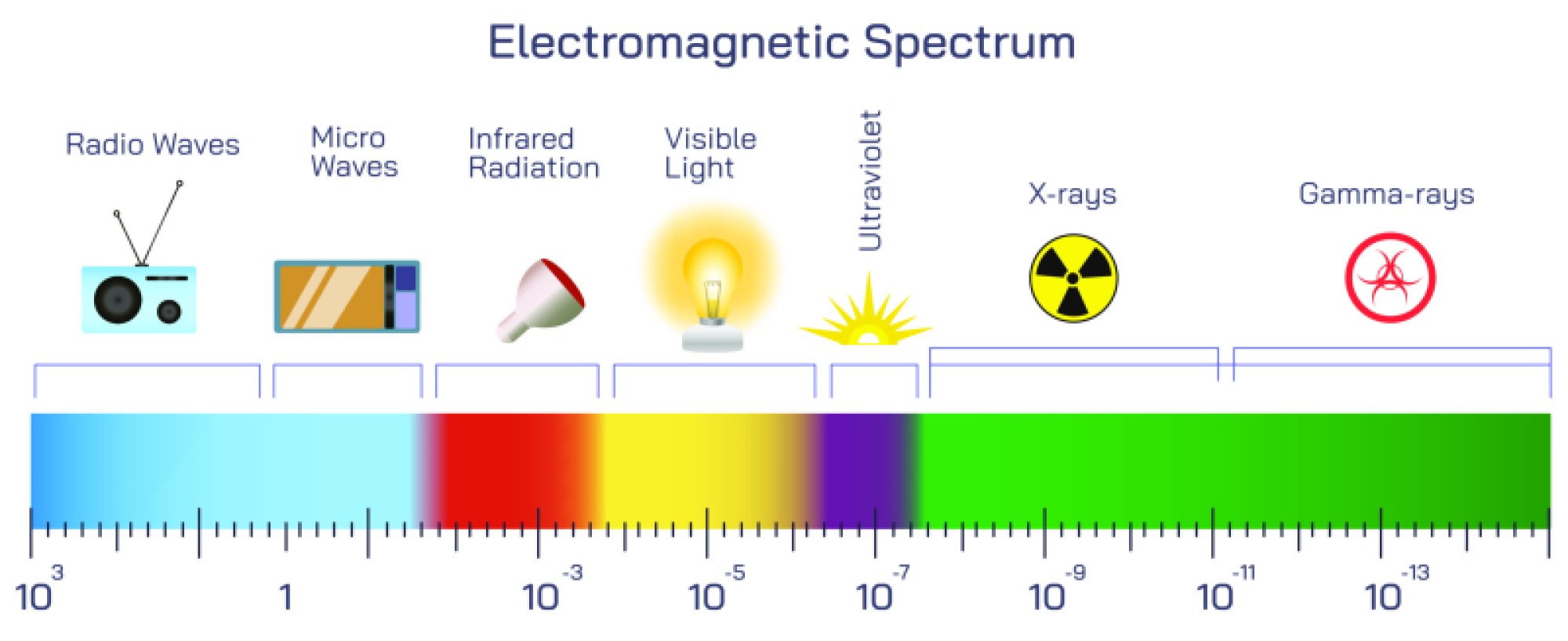
The illustration usually begins with the macroscopic water droplet, recognizable as a smooth, cohesive bead of liquid. This stage highlights the familiar physical properties of water—surface tension, fluidity, droplet shape, and the way water interacts with light. Labels often emphasize hydrogen bonding networks that give water a high level of cohesion, enabling a drop to maintain its curved surface. Even at this outermost scale, the behavior of the droplet is determined by interactions among billions upon billions of particles.
The image then transitions to the microscopic realm, revealing the densely packed network of water molecules (H₂O). Each molecule consists of two hydrogen atoms bonded to one oxygen atom. A typical illustration shows the well-known “V shape” of the water molecule with an angle of about 104.5°, emphasizing the asymmetric placement of atoms. Partial charges—slightly negative around oxygen and slightly positive around hydrogen—are visualized to explain why water molecules attract each other strongly through hydrogen bonds. At this molecular zoom level, the droplet transforms into a swirling network of constantly moving units rather than a smooth, continuous mass.
A further zoom reveals the individual atoms within the water molecule. Here the oxygen and hydrogen atoms appear as distinct spheres or electron-density shells, signifying that each atom is itself a composite structure. Oxygen contains eight protons, eight neutrons, and eight electrons; hydrogen contains one proton and one electron. Illustrations typically show electrons occupying orbitals surrounding the nucleus rather than orbiting like planets, highlighting the probabilistic nature of quantum electron clouds. At this scale, chemistry dominates—atoms bond by sharing electrons, forming the stable O–H bonds that give the molecule its geometry.
Following the atomic level, the illustration zooms into the atomic nucleus, where protons and neutrons cluster tightly together through the strong nuclear force. Diagrams of this region often depict a dense core resembling a cluster of spheres locked together. The nucleus appears extremely tiny even compared to the atom—if an atom were the size of a football stadium, the nucleus would be smaller than a grain of rice. This jump in scale makes clear just how much empty space exists within matter. Protons and neutrons inside this nucleus each contribute almost all of the atom’s mass, although they occupy only a tiny fraction of its volume.
Once the viewpoint enters a proton or neutron, the illustration arrives at the subatomic particle level. In modern particle physics, protons and neutrons are not elementary. They are composed of smaller constituents called quarks, which are held together by force-carrying particles called gluons. A detailed zoom typically portrays three quarks interacting dynamically inside each proton or neutron. In a proton, two up quarks and one down quark appear; in a neutron, the configuration is one up quark and two down quarks. Gluons are shown as flowing bonds or spring-like connections, continuously exchanged to bind the quarks with the strong nuclear force. At this level, matter no longer resembles solid structures but becomes a vibrating sea of quantum fields, where particles blink into and out of existence moment by moment.
Some illustrations extend even deeper, hinting at quantum fields or string-like foundations. Although still theoretical, string-inspired visuals represent quarks not as tiny beads but as oscillating filaments of energy, suggesting that fundamental matter arises from patterns of vibration rather than rigid miniature particles. This final optional scale layer emphasizes the idea that the most fundamental elements of a water droplet might be more like dynamic energy excitations than material lumps.
By linking each scale into the next, a molecule-to-quark illustration shows a striking contrast in both size and conceptual framework:
• The macroscopic droplet is governed by gravity, cohesion, and fluid mechanics.
• Molecules and atoms are governed by chemical bonding and quantum electron interactions.
• Nuclei are held together by nuclear forces far stronger than chemical ones.
• Quarks interact through the strong force and quantum chromodynamics, the most fundamental theory currently describing matter.
Even more profoundly, the amount of material within each level changes in unimaginable magnitude. A one-millimeter water droplet contains roughly 10²¹ molecules; the number of atoms and subatomic particles multiplies even further. This highlights that the everyday world is constructed from a staggering number of components whose collective behavior yields the liquid form we experience.
In the final view, the illustration bridges the scales visually: a water droplet becoming a cluster of molecules, shrinking into atoms, condensing into nuclei, deepening into protons and neutrons, and finally dissolving into quarks bound by gluon fields. Despite the enormous differences in scale—spanning more than 20 orders of magnitude—each level is essential to the next. The formation of a water droplet depends on hydrogen bonds between molecules; hydrogen bonds depend on shared electrons; electrons orbit nuclei stabilized by protons and neutrons; and those nuclear particles exist only because quarks are confined and energized by the strong force.
In this way, the illustration transforms a drop of water from a familiar fluid into a layered structure of extraordinary depth. It reveals that something as ordinary as a raindrop is built from the same quantum particles that compose stars, planets, and life. By visually descending from the macroscopic to the subatomic, the diagram tells a seamless scientific story: the universe is consistent across scale, and every ordinary object hides a universe of structure within it.