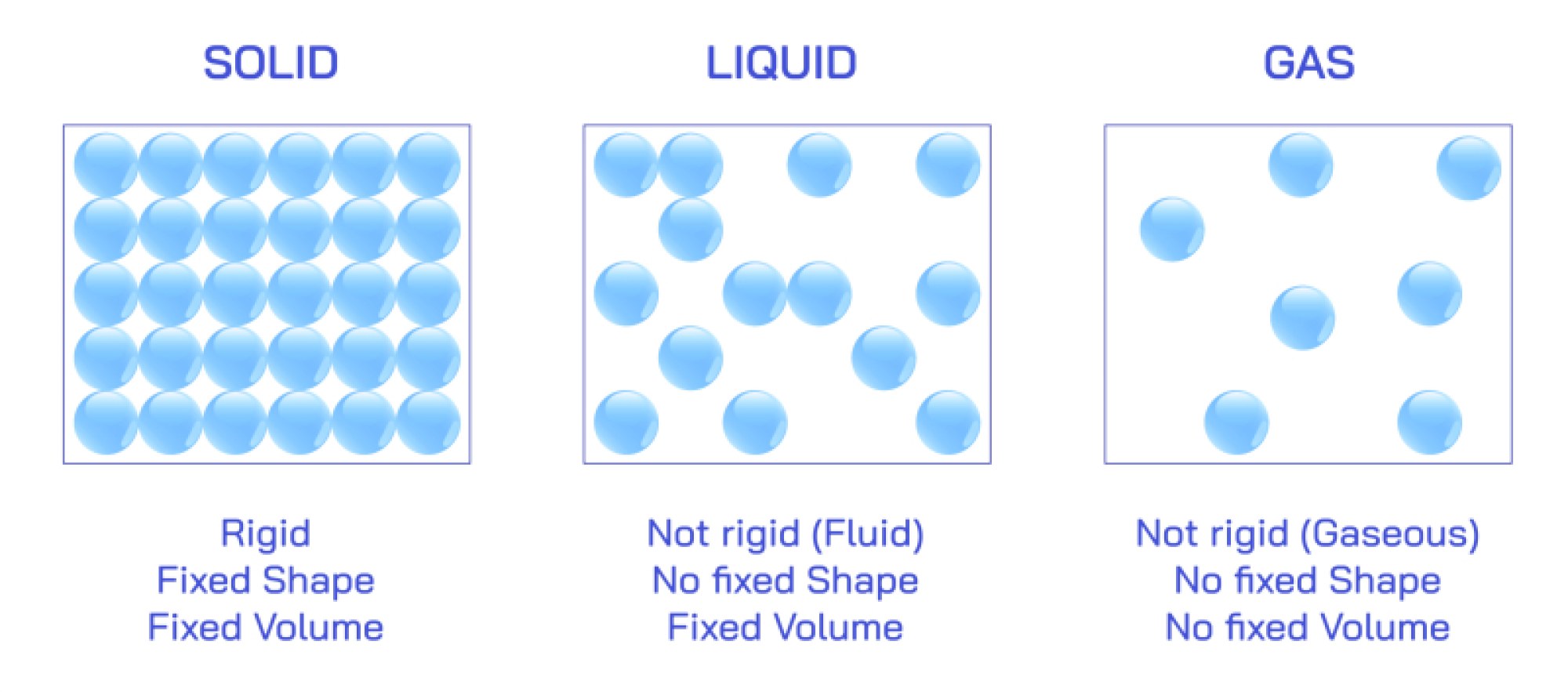
Copper Element Illustration: Atomic Structure, Properties, and Applications Explained
Copper is one of the most significant and recognizable elements in human civilization, and an illustration that highlights its atomic structure, physical properties, and countless applications helps explain why this element has shaped technological, industrial, cultural, and economic development for thousands of years. On the periodic table, copper is represented by the symbol Cu and holds atomic number 29, which means that each copper atom contains 29 protons in its nucleus, balanced by 29 electrons arranged around it. When drawn in an atomic diagram, copper’s electron configuration is shown as 2 electrons in the first shell, 8 in the second, 18 in the third, and 1 in the fourth outer shell, giving copper a single valence electron. This lone outer electron is only loosely held, allowing it to move freely when copper atoms are packed in a metallic lattice. The illustration of this electron mobility becomes the key to understanding why copper is such an excellent electrical and thermal conductor. In a cluster of copper atoms shown bonded together in solid form, the outermost electrons wander fluidly among the atoms rather than remaining tied to a single one, forming an “electron sea” that enables rapid transmission of electrical charge and heat through the metallic lattice.
In more detailed structural illustrations, copper atoms are arranged in a face-centered cubic (FCC) lattice. Each copper atom sits at the corners and center of each face of a cube, and this arrangement creates a dense, highly symmetrical structure that enhances strength, ductility, and malleability. Because the atoms are closely packed but able to slide over each other without breaking lattice integrity, copper can be stretched into wires, hammered into sheets, or bent without fracturing, making it exceptionally workable. Illustrations of the FCC lattice usually show how the tightly packed planes allow dislocations to move easily through the lattice, accounting for copper’s formability. This same structure also contributes to copper’s excellent resistance to mechanical fatigue, explaining why copper remains reliable in applications subject to vibration or repeated motion such as machinery, motors, transformers, and electrical connectors.
Visually, copper is notable for its distinctive reddish-orange metallic luster, which few other metals naturally display. In a chemical illustration of copper’s electron arrangement, this color originates from transitions between energy levels that selectively absorb light wavelengths in the blue region of the visible spectrum, reflecting reddish-orange wavelengths back to the observer. This characteristic shine is one reason copper has long been used decoratively in jewelry, sculpture, architecture, metalwork, and coinage. As copper is exposed to oxygen and moisture over time, the surface undergoes a process known as patination, in which a thin protective coating forms—most famously the blue-green patina of copper carbonate and copper sulfate, seen on rooftops, domes, and iconic structures like the Statue of Liberty. Illustrations showing the pristine copper surface gradually developing through oxidized brown phases and into the ultimately stable green patina help viewers understand that this transformation is not decay but protection; the patina prevents further corrosion by sealing the underlying metal from environmental contact.
In terms of mechanical and physical properties, copper is typically illustrated in learning materials with comparative graphs showing its high electrical conductivity, high thermal conductivity, excellent corrosion resistance, high ductility, and moderate tensile strength. Side-by-side bar charts often show copper surpassing most metals in conductivity except for silver, but because copper is far more abundant and cost-effective, it became the world’s primary conductive metal. For clarity, illustrations may show copper wires transferring electricity within a circuit and copper coils in motors dissipating heat efficiently. Another property often included in diagrams is copper’s non-magnetic character, which makes it valuable in electromagnetic shielding and in precision instruments requiring materials that do not interfere with magnetic fields.
Copper’s remarkable behavior in alloy formation is another important feature highlighted in scientific illustrations. When copper is combined with other metals, the resulting alloys demonstrate enhanced and specialized properties. Copper and tin form bronze, one of the earliest engineered materials in human history, marking the Bronze Age and revolutionizing tools, weapons, and cultural development. Copper and zinc form brass, known for its bright golden appearance, corrosion resistance, antimicrobial qualities, and ease of machining, making it indispensable for musical instruments, plumbing fixtures, fittings, and decorative objects. Copper-nickel alloys are used in naval applications because of their resistance to seawater corrosion. Illustrations of alloy formation typically depict atomic mixtures showing how secondary metal atoms fit into copper’s lattice, altering its crystal structure and enhancing mechanical or chemical properties depending on composition. These illustrations reinforce the idea that copper is both useful in pure form and extraordinarily adaptable through alloying.
The role of copper in electrical infrastructure is easy to depict and profoundly important. In a cross-section illustration of a household or industrial wiring system, copper strands appear forming the conductive core of electrical cables that power everything from lighting and appliances to telecommunications. In motors and generators, copper coils wrapped around iron cores create magnetic fields that convert electrical energy to mechanical motion or vice versa. In transformers, copper windings channel alternating current while dissipating heat more efficiently than most other metals. In electronics, copper pathways etched onto printed circuit boards move signals through microprocessors and memory systems. In renewable energy technology, copper is integral to wind turbines, solar panels, and electric vehicle motors. Illustrations of modern energy grids often highlight copper connecting power generation, transmission, distribution, and consumption, signifying that electricity is not useful without a conductor capable of channeling it safely and efficiently.
Copper is also crucial in plumbing and heating systems, and illustrations commonly show copper pipes carrying water in residential and commercial buildings. The reason for copper’s dominance in plumbing is both chemical and mechanical: copper does not rust, it withstands temperature changes, and it naturally resists microbial growth. This antimicrobial property is powerful enough that copper surfaces in hospitals and public facilities reduce transmission of harmful microorganisms. In diagrams of the respiratory and bacterial cell membrane interaction, copper ions penetrate and destabilize microbial cell walls, disrupt proteins, and damage genetic material. These diagrams reinforce the emerging public health applications of copper in touch surfaces, medical equipment, and sterilization systems.
Beyond technology and infrastructure, copper plays important roles in biology and human health. Although humans need only small quantities, copper is an essential trace mineral required for enzymes involved in energy production, connective tissue formation, and iron metabolism. Illustrations of biological copper often show atoms bonded within cytochrome c oxidase, a central enzyme in the mitochondrial electron transport chain, demonstrating copper’s contribution to cellular respiration. In the bloodstream, a balance of copper supports red blood cell development, immune system function, and neurological health. In plants, copper appears in illustrations showing its involvement in chlorophyll formation and enzyme systems; deficiencies lead to stunted growth or loss of pigmentation, whereas excessive copper becomes toxic. These biological illustrations reinforce that copper is not merely a structural and industrial metal but an element woven into living systems.
Copper’s industrial applications extend further into heat transfer systems, semiconductor devices, machinery components, art and architecture, transportation, and telecommunications. Heat exchangers rely on copper’s thermal conductivity to move heat rapidly between fluids. Electric vehicles and charging infrastructure require copper in quantities higher than traditional automobiles. Semiconductor manufacturing uses copper interconnects inside microchips to boost speed and efficiency. Illustrations that follow copper from ore extraction to refining, alloy production, and final applications portray the metal as one of the most economically significant materials of modern life. The copper cycle—beginning with mining of ores such as chalcopyrite, smelting and electrorefining, manufacturing, usage, recycling, and reuse—is often displayed to emphasize sustainability. Copper is highly recyclable and retains its quality indefinitely, making it one of the most environmentally valuable industrial metals.
The connection between copper and human history is another theme often included in educational illustrations. Ancient civilizations such as Mesopotamia, Egypt, the Indus Valley culture, and Greece used copper for weapons, coins, jewelry, and tools. The transition from stone to metal in human development is symbolically represented by the widespread adoption of bronze, a copper alloy that enabled advances in agriculture, architecture, warfare, and trade. Maps and artifacts shown alongside copper deposits demonstrate how access to copper resources influenced the rise of civilizations, migration routes, and early global commerce long before the modern era.
Today, copper remains indispensable because of a rare combination of properties: it conducts electricity and heat with exceptional efficiency, resists corrosion, forms strong and adaptable alloys, provides antimicrobial capability, and remains workable while retaining strength. Illustrations summarizing these qualities typically place copper at the center of a diagram surrounded by its functional roles—energy systems, electronics, construction, climate technologies, health, and manufacturing—revealing how deeply this element is embedded in the functioning of modern society.
Ultimately, an illustration of copper’s atomic structure, physical characteristics, and applications provides more than visual clarity—it explains why this single element stands at the crossroads of chemistry, engineering, biology, technology, and culture. The lone electron in its outer shell, the symmetry of its crystal lattice, the shine of its surface, and the stability of its patina together hint at copper’s broader narrative: an element uniquely positioned to serve as a foundation for both ancient craftsmanship and cutting-edge innovation. Copper embodies the idea that our world is built not only from numbers, machines, and circuits, but from the behavior of atoms—one of which has guided and empowered human progress from the earliest metal blades to the most sophisticated renewable energy systems of the future.