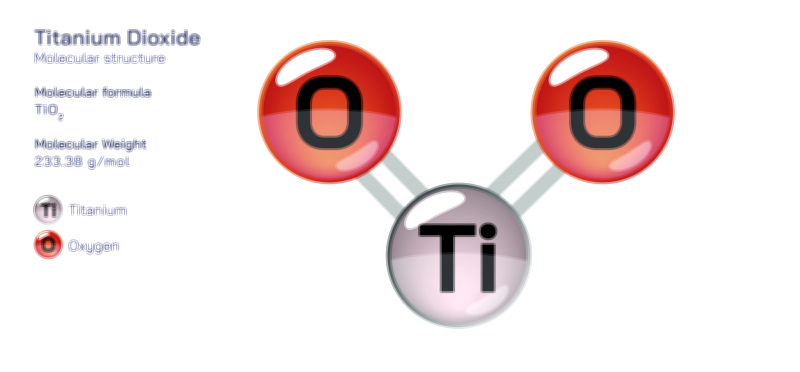
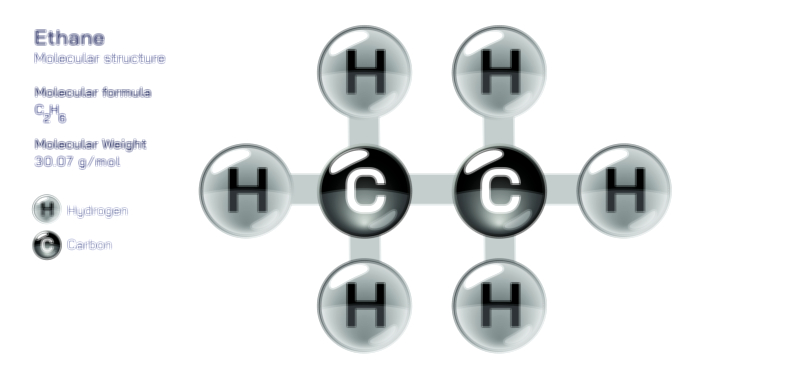
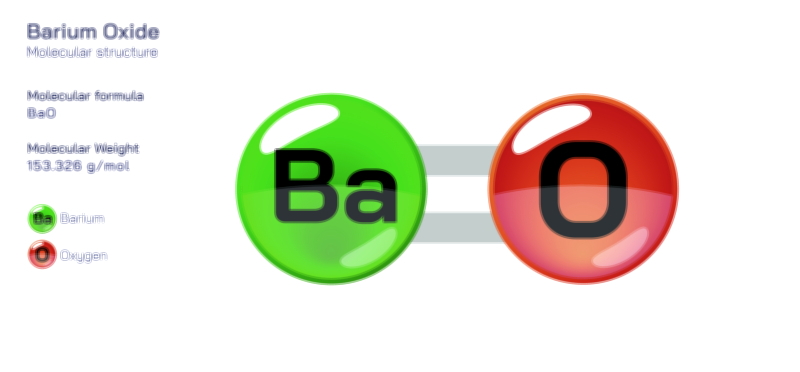
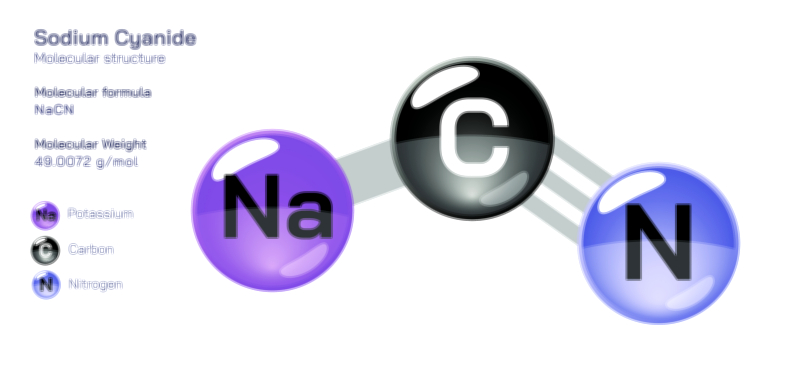
ØÉÆØÉóØÉźØÉóØÉ£ØÉ©Øɦ ØÉāØÉóØÉ©ØÉ▒ØÉóØÉØØÉ× ŌĆö ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ× ØÉÜØɦØÉØ ØÉīØÉÜØÉŁØÉ×ØɽØÉóØÉÜØÉź ØÉéØÉĪØÉÜØɽØÉÜØÉ£ØÉŁØÉ×ØɽØÉóØɼØÉŁØÉóØÉ£Øɼ (ØÉäØÉØØÉ«ØÉ£ØÉÜØÉŁØÉóØÉ©ØɦØÉÜØÉź ØÉŗØÉ©ØɦØÉĀ ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓)
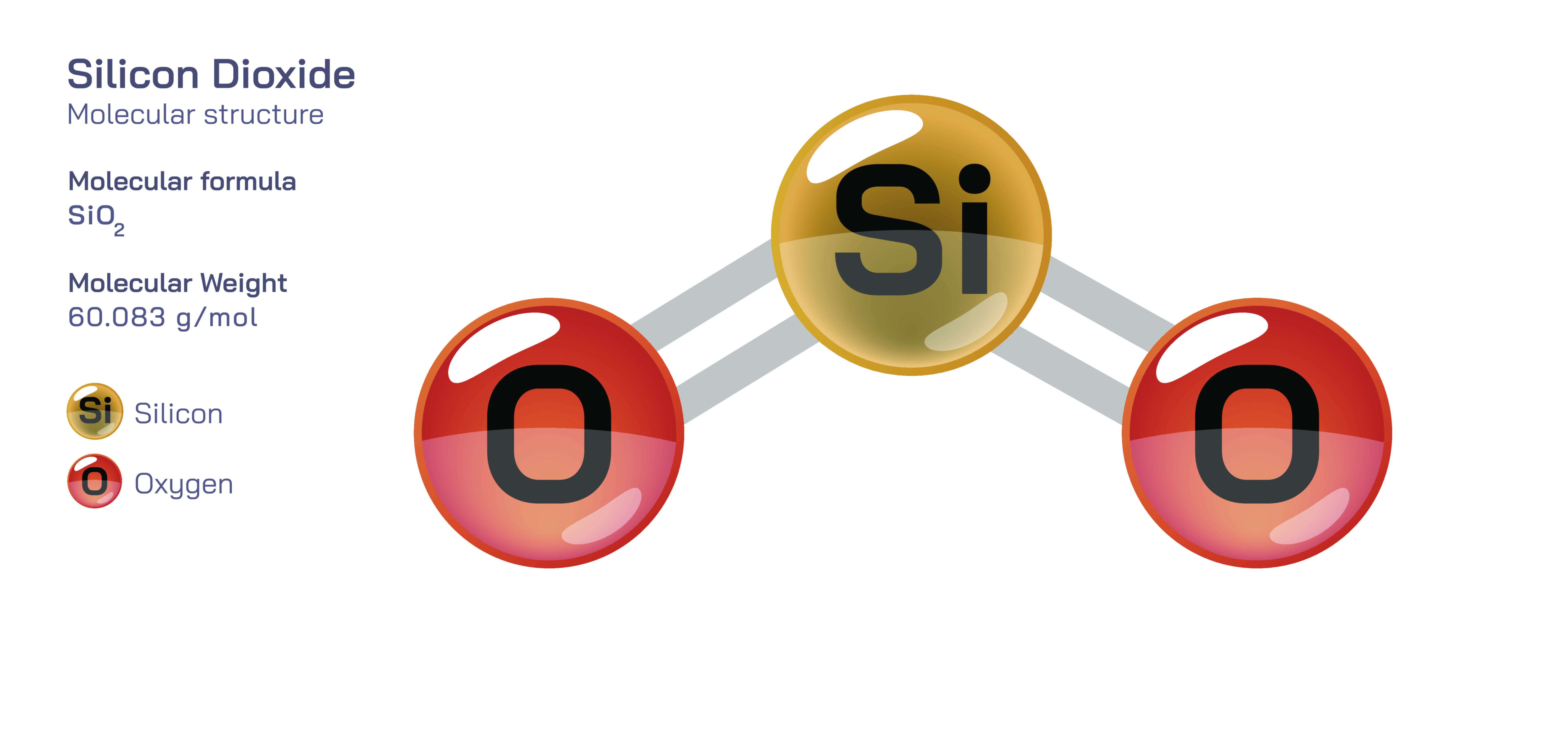
Silicon dioxide, commonly referred to as SiOŌéé, is one of the most abundant and structurally influential compounds on Earth, forming the primary component of sand, quartz, glass and numerous mineral forms. Although visually simple in its raw natural state, silicon dioxide is built from a highly ordered atomic arrangement that determines its exceptional physical strength, chemical stability and role in both geological and technological processes. The compound consists of silicon and oxygen bonded together in a ratio of one silicon atom to two oxygen atoms, yet the way these atoms are linked extends far beyond a single SiOŌéé unit. Instead of existing as isolated molecules, silicon dioxide forms continuous networks of siliconŌĆōoxygen tetrahedra connected through shared oxygen atoms, creating rigid three-dimensional frameworks. This connectivity gives SiOŌéé its well-known hardness and resistance to heat, weathering and chemical attack, placing it among the most durable naturally occurring materials in the EarthŌĆÖs crust.
The structural foundation of silicon dioxide lies in the siliconŌĆōoxygen tetrahedron: a silicon atom at the centre linked covalently to four oxygen atoms positioned at the corners of a tetrahedral shape. Each oxygen atom attaches not solely to the central silicon but also bridges to adjacent silicon atoms, which produces an extended network rather than discrete molecules. As a result, the empirical formula SiOŌéé does not describe individual molecules but rather the ratio of atoms in a vast covalent lattice. The strength of the bonds in this tetrahedral framework accounts for SiOŌééŌĆÖs extremely high melting temperature and its role in forming minerals that withstand erosion and geological transformation over millions of years. In quartz, the tetrahedra are arranged in a repeating crystalline pattern, whereas in amorphous silicaŌĆösuch as glassŌĆöthe tetrahedra are still connected, but without long-range order. This distinction between crystalline and amorphous silica demonstrates how the same chemical formula can create different physical materials depending on atomic organization.
Silicon dioxide also holds immense technological significance because of its unique electrical and optical properties. In the semiconductor industry, SiOŌéé acts as an insulating material and a gate dielectric in silicon-based microchips. When thermally grown on silicon wafers, it forms extremely thin, uniform layers that trap and control electrical charge, a principle crucial for designing computer processors, memory storage devices and integrated circuitry. The optical transparency of amorphous silica across the visible and infrared spectrum is foundational to the production of glass, optical fibres, lenses and laboratory equipment. In fibre-optic communication systems, silicaŌĆÖs low optical loss allows light to travel long distances with minimal weakening, enabling global high-speed data transmission. The same structural rigidity that strengthens quartz also contributes to the low thermal expansion and high temperature tolerance of fused silica, which is used in scientific instruments, satellites and high-intensity lighting.
Beyond geological and technological applications, silicon dioxide demonstrates versatility in chemical, environmental and biological contexts. It plays an important role in soil composition and nutrient retention, supporting plant growth by regulating mineral balance and moisture distribution. In food and pharmaceutical industries, finely powdered amorphous silica is used as an anti-caking agent, stabilizer and flow regulator due to its surface adsorption properties. At the microscopic scale, silica particles provide large surface area and tunable porosity that make them valuable in chromatography, catalysis and controlled drug-delivery systems. Even in consumer products, SiOŌéé appears in toothpaste for its polishing ability and in cosmetics for its light-diffusing properties.
Although silicon dioxide is generally regarded as inert and non-toxic, its behaviour depends on particle size and physical form. Inhalation of crystalline silica dust over prolonged periods can cause lung diseases such as silicosis because the sharp rigid particles trigger inflammation in respiratory tissue. Conversely, food-grade amorphous silica is biologically compatible and safely excreted by the body. This contrast highlights that chemical safety depends not only on composition but also on material structure, particle morphology and exposure environment.
Silicon dioxide therefore embodies a compound whose structural architecture governs its function in the natural world and in human innovation. From building mountains and beaches to powering telecommunications and semiconductor electronics, SiOŌéé demonstrates how atomic geometry transforms a simple formula into one of the most essential materials in history. The tetrahedral bonding network defines its identity, proving that the true nature of a substance lies not only in the atoms present, but in how those atoms are arranged and connected across microscopic space.