Vitamin C Structure — Understanding the Ascorbic Acid Compound
The Essential Nature of Vitamin C as a Structural Compound
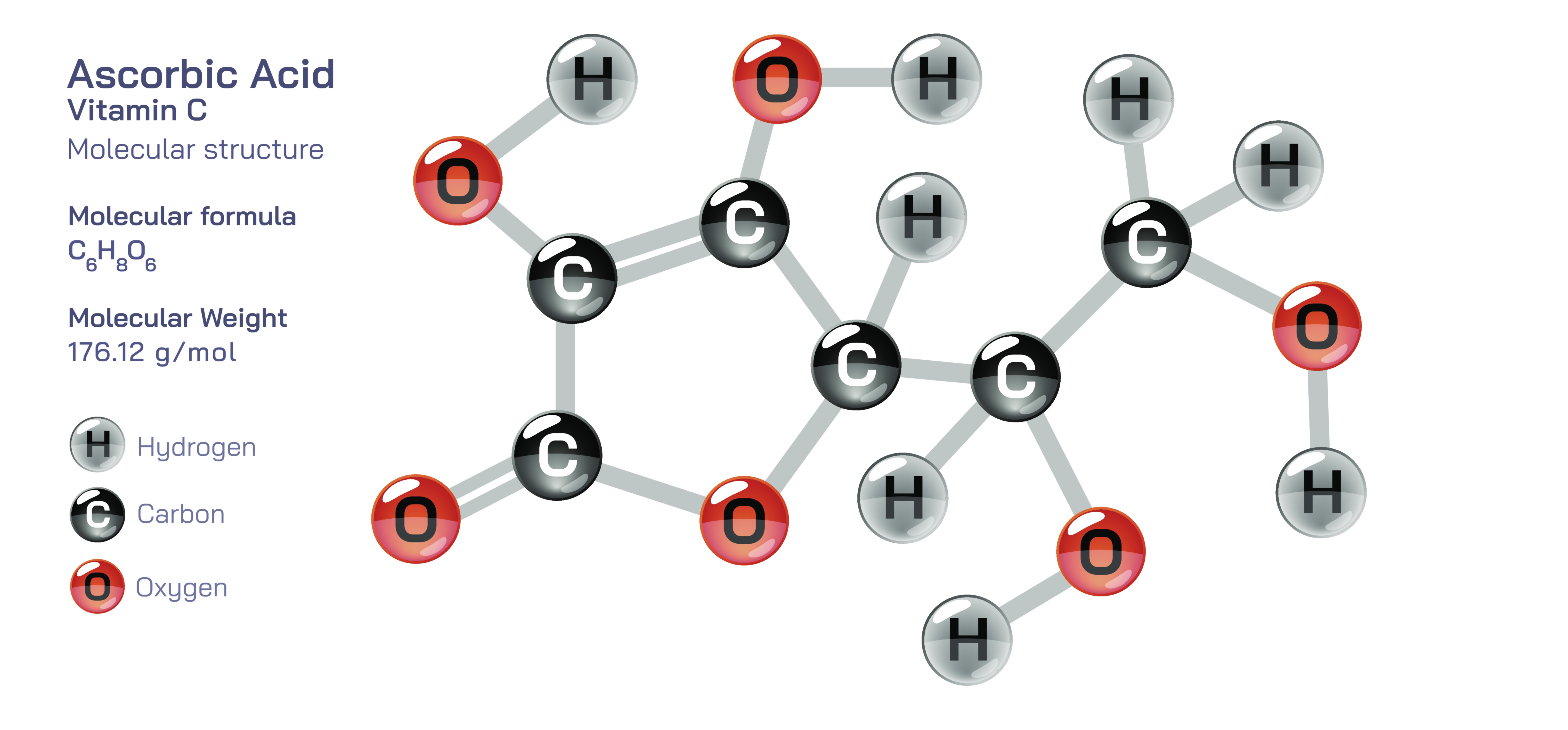
Vitamin C, chemically known as ascorbic acid, is a small but highly active organic compound whose molecular structure enables it to support numerous vital functions within the human body. Its structure is built around a compact ring made of carbon and oxygen atoms, forming a framework that gives the molecule both stability and reactivity. This ring is surrounded by several groups that interact easily with water, which is why vitamin C dissolves quickly in bodily fluids and circulates efficiently through the bloodstream. Because it is water-soluble, the body cannot store large reserves of it, making regular dietary intake essential. The structural arrangement of vitamin C is what allows it to act as a powerful antioxidant, a collagen-building agent, and a key player in immune defense. Its design reflects a balance between strength and flexibility, enabling it to participate in countless biochemical reactions without losing its effectiveness.
How the Structure Supports Antioxidant Protection and Stability
The molecular structure of ascorbic acid contains specific reactive groups that allow it to donate protective electrons to unstable molecules in the body. These unstable molecules naturally form during metabolism, stress, pollution exposure, and illness, and they can damage tissues if they remain unchecked. Vitamin C’s structure makes it incredibly efficient at neutralizing these harmful molecules, safeguarding cells from internal stress. The unique arrangement of its atoms allows vitamin C to shift smoothly between its active and stabilized forms without becoming harmful itself. This is why it is considered one of the safest and most effective antioxidants. In structural illustrations, vitamin C is often shown with its ring-shaped skeleton and distinct reactive features, emphasizing how its chemical layout contributes directly to its protective role.
The Structure Behind Collagen Formation and Tissue Repair
Vitamin C’s molecular architecture also enables it to support one of the body’s most essential functions: the formation and strengthening of collagen. Collagen is a structural fiber found in skin, bones, blood vessels, and connective tissues. Specialized enzymes in the body depend on vitamin C’s structural reactivity to stabilize these fibers during formation. Without vitamin C, collagen becomes weak and poorly formed, leading to fragile skin, slow wound healing, and weakened blood vessels. The compound’s ability to participate in these specific enzymatic reactions is deeply rooted in the arrangement of its functional groups, which interact optimally with the enzymes responsible for collagen assembly. This is why deficiencies in vitamin C can result in visible physical complications and why the molecule remains essential for maintaining structural integrity throughout the body.
How Vitamin C’s Structure Supports Nutrient Absorption and Metabolism
Another important aspect of vitamin C’s structure is its ability to assist in nutrient absorption, particularly iron from plant-based sources. When vitamin C encounters dietary iron in the digestive system, its reactive structure helps convert the mineral into a form that the body can absorb more effectively. This protective interaction prevents iron from forming unusable complexes and enhances overall metabolic efficiency. The molecule’s water-soluble and reactive design allows it to move easily between digestive tissues and the bloodstream, supporting these conversions with remarkable efficiency. Its structural flexibility also enables it to participate in detoxification processes and metabolic pathways that depend on quick chemical exchanges. These qualities highlight how the compound’s molecular shape determines its broad influence on nutrition and metabolism.
Why Vitamin C Is Sensitive to Environmental Conditions
The same structural features that make vitamin C reactive and beneficial also make it vulnerable to external factors like heat, oxygen, and prolonged exposure to light. When vitamin C encounters these conditions, the delicate arrangement of its reactive groups begins to break down, reducing its potency. This is why fresh fruits and vegetables—particularly citrus fruits, peppers, berries, and leafy greens—are important dietary sources, as they contain vitamin C in its most active form. Cooking, storage, and exposure to air gradually weaken its structure. The sensitivity of vitamin C’s molecular design underscores the importance of consuming fresh foods and highlights why supplements and fortified products must be stored carefully to preserve their effectiveness.
The Relationship Between Structure and Immune Support
Vitamin C’s structure allows it to support the immune system in multiple ways. Immune cells depend heavily on antioxidants to function under stressful conditions, especially during infections or inflammation. Vitamin C accumulates inside these cells, using its reactive groups to protect them from internal damage and support their activity. It helps stimulate the production and performance of several immune components, allowing the body to respond quickly and efficiently to threats. Because of its compact, water-friendly structure, vitamin C travels rapidly to areas of the body where immune activity is highest. Its ability to cycle between active and stabilized forms gives it lasting influence during periods of intense immune demand.
Why Understanding Vitamin C’s Structure Enhances Its Importance
The molecular structure of ascorbic acid is the foundation of its effectiveness. Its ring-like shape, reactive edges, and water-soluble design are responsible for its diverse roles—from antioxidant protection and tissue repair to metabolic support and immune strength. Understanding its structure reveals why the body relies on vitamin C so heavily and why consistent intake is necessary. It also highlights why vitamin C is considered essential rather than optional: its molecular design enables it to act in ways no other nutrient can fully replace. Appreciating the architecture of this small compound deepens understanding of its enormous impact on daily health, longevity, and resilience.