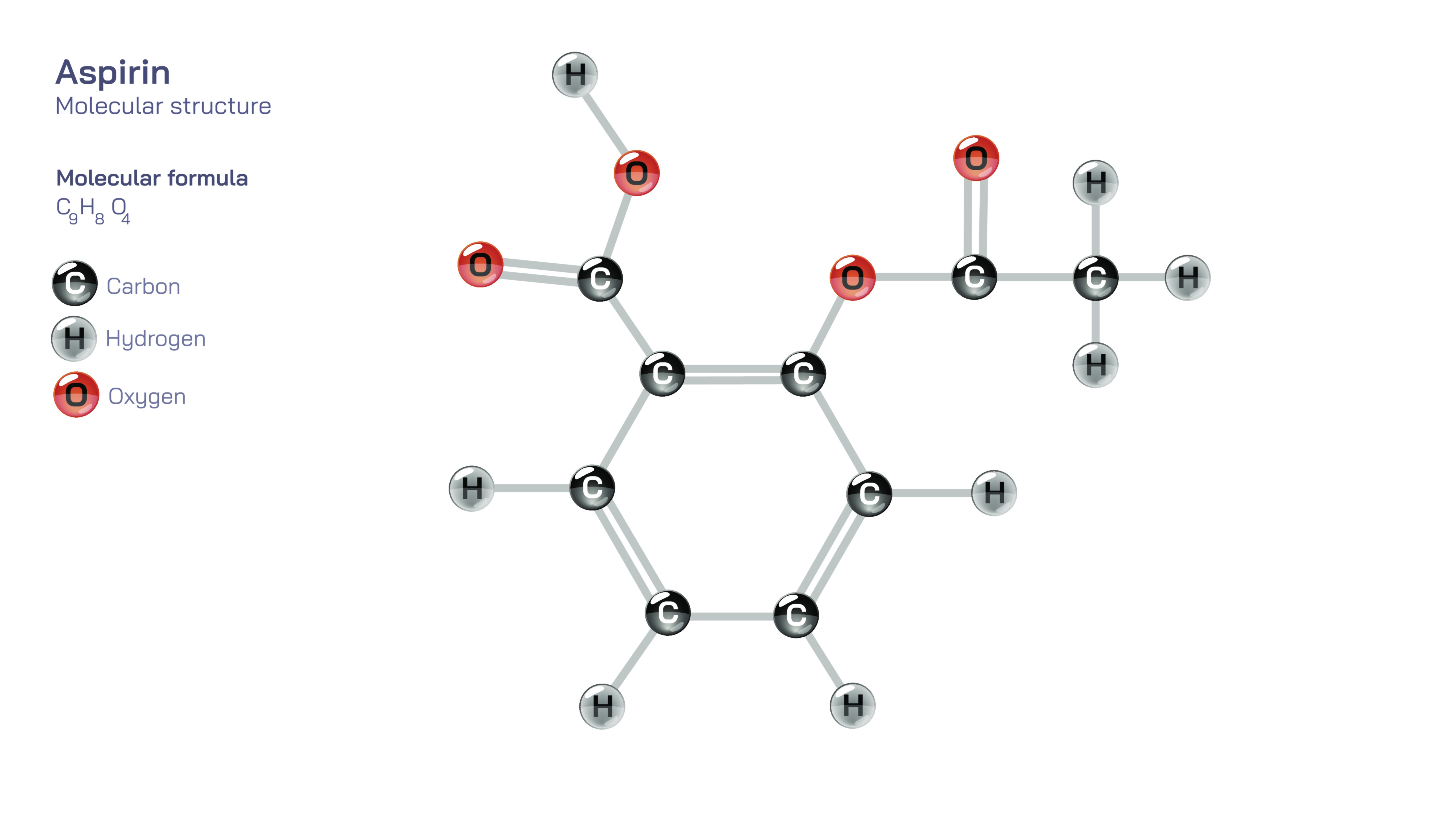
Aspirin — Understanding the Molecular Structure of Acetylsalicylic Acid
The Essential Nature of Aspirin as a Structured Organic Compound
Aspirin, known scientifically as acetylsalicylic acid, is one of the most widely used medical compounds in the world, appreciated for its pain-relieving, fever-reducing, and anti-inflammatory effects. At the center of its effectiveness lies its molecular structure, which is based on a compact arrangement of carbon, hydrogen, and oxygen atoms forming a stable aromatic ring connected to reactive functional groups. This structure gives aspirin the unique ability to interact with key biological pathways that regulate inflammation and pain. The aromatic ring provides strength and rigidity, while the attached acetyl group transforms it from a simple natural compound into a powerful therapeutic agent. This structural enhancement is what allows aspirin to influence the body’s chemical signals so efficiently, making it one of the most enduring and trusted medications in modern healthcare.
How the Arrangement of Atoms Defines Aspirin’s Activity
Aspirin’s structure includes a core ring that anchors the molecule and two reactive extensions that determine its behavior inside the body. One part of the molecule is related to salicylic acid, a compound originally derived from willow bark and known for soothing discomfort. The addition of the acetyl group is what gives aspirin its name and its enhanced therapeutic function. This acetyl portion is highly reactive and attaches firmly to specific enzymes in the body, altering their activity in a way that reduces inflammation and pain. This interaction is only possible because of the precise orientation of atoms in the aspirin molecule. In illustrations of its structure, the molecule is often shown with its aromatic ring and branching groups, highlighting the distinctive arrangement that enables aspirin to intervene in biological signaling.
How Aspirin’s Structure Influences Pain Relief and Inflammation Control
The powerful effect of aspirin comes from its ability to calm the body’s inflammatory responses at a chemical level. When tissues become irritated or damaged, the body produces certain signaling substances that create sensations of pain, heat, swelling, and discomfort. Aspirin’s structure allows it to attach to the enzymes responsible for creating these substances, effectively slowing down their formation. This attachment is made possible by the function of the acetyl group, which interacts precisely with the enzyme’s active region. As aspirin binds, it gently shifts the enzyme’s behavior, reducing its ability to produce pain-related signals. This gentle but effective molecular interaction helps restore comfort, ease tension, and reduce inflammation. Without its specific structural features, aspirin would not have the same therapeutic influence on these natural pathways.
The Molecular Design Behind Aspirin’s Versatility
Aspirin’s structure also enables it to have broader effects beyond pain and inflammation relief. Its interaction with chemical regulators in the bloodstream gives it an additional ability to reduce the formation of internal clots in certain individuals. This is because its molecular design allows it to influence signals that normally make platelets—a type of blood cell—stick together. By moderating these signals, aspirin supports smoother blood flow, which is why it may be used in low doses for long-term cardiovascular protection. This secondary benefit is directly tied to the molecule’s acetyl-bearing structure, which gives it the flexibility to interact with different systems inside the body. The same features that allow it to act on inflammatory pathways also make it capable of influencing blood-related processes, highlighting the remarkable efficiency of its design.
Why Aspirin’s Structure Makes It Effective but Also Requires Care
The reactive portions of the aspirin molecule that provide strong benefits also require thoughtful use, because they can interact with tissues such as the stomach lining. Aspirin’s molecular shape allows it to act quickly once it enters the bloodstream, but before it reaches circulation, it must pass through the digestive system. Because its functional groups are active, they can occasionally irritate sensitive tissues when taken in high doses or without appropriate protection. This is why modern formulations and medical guidance focus on balancing aspirin’s benefits with safe usage practices. The molecule’s design is powerful, but like many effective compounds, its reactivity must be managed carefully to ensure it supports the body rather than overwhelming it.
Environmental Sensitivity and Stability of Acetylsalicylic Acid
Aspirin’s structure, while strong enough to perform reliably inside the body, is sensitive to certain environmental conditions. Exposure to moisture or prolonged storage in humid environments can gradually break down the acetyl group, reducing the effectiveness of the medication. The molecule is designed to remain stable under normal conditions, but its reactive nature means it should be stored properly to preserve its potency. Illustrations of the molecule often highlight the reactive portion that is most vulnerable to environmental changes. This characteristic is a natural part of its design, reflecting the balance between structural stability and the reactivity needed for therapeutic action.
How Understanding the Structure Deepens Appreciation of Its Benefits
The molecular structure of aspirin may appear small and simple, but it contains a remarkable level of precision and purpose. Each part of the molecule contributes to its broad therapeutic effects, from pain relief and fever reduction to cardiovascular support. Understanding how its structure is arranged helps reveal why aspirin has remained such an important medication for more than a century. Its stability, reactivity, and versatility all stem from the thoughtful arrangement of its atoms. By appreciating this structure, one gains a deeper understanding of how a single well-designed compound can support health, ease discomfort, and protect the body from internal stresses.