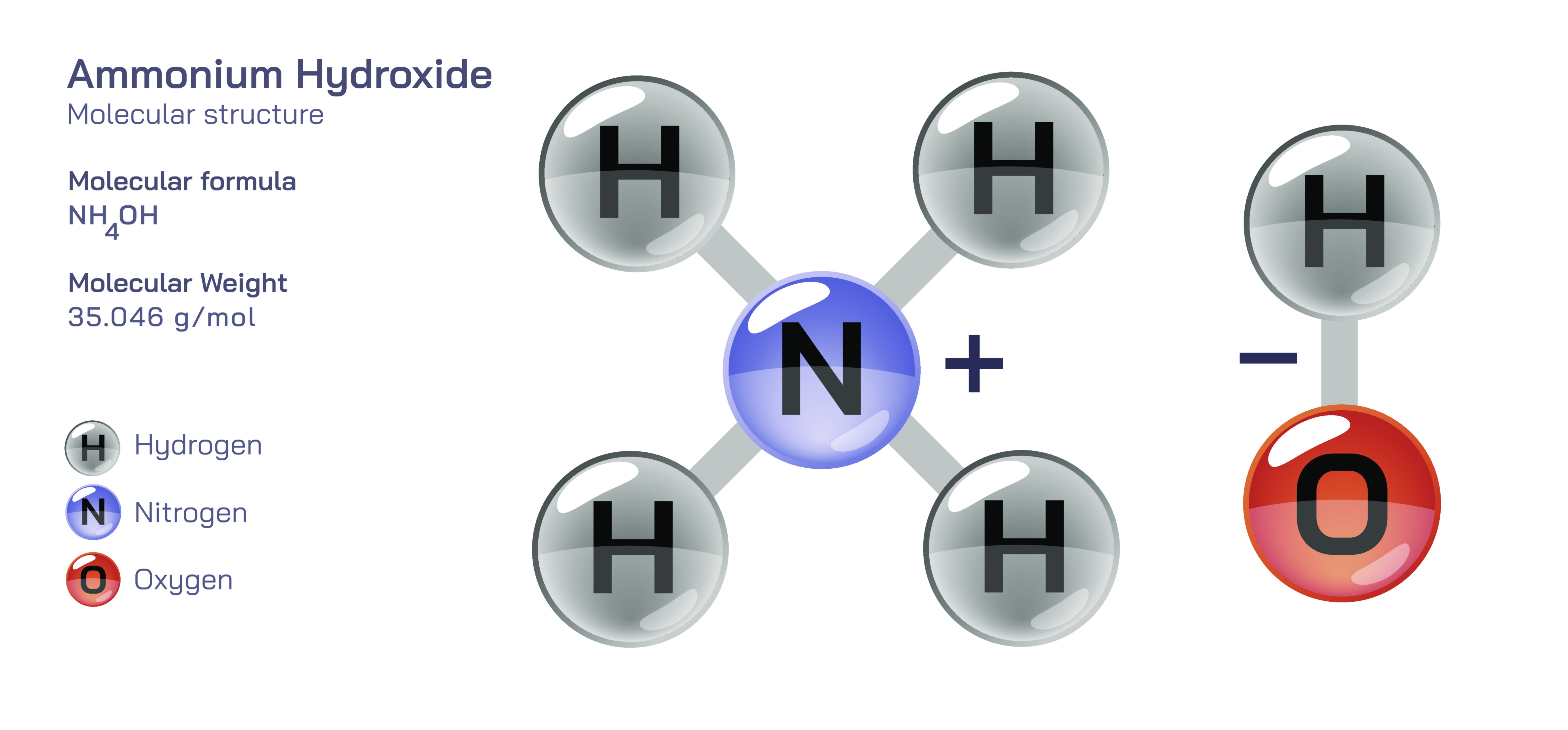
Ammonium Hydroxide — Chemical Solution Structure, Properties, and Behavior
Understanding the True Nature of Ammonium Hydroxide
Ammonium hydroxide is widely recognized as a basic chemical solution, yet its true nature is often misunderstood. Rather than being a standalone, isolatable compound, ammonium hydroxide is best described as ammonia dissolved in water. When ammonia gas comes into contact with liquid water, a portion of its molecules interact with water molecules, forming ammonium ions and hydroxide ions. However, the solution does not contain distinct “ammonium hydroxide molecules”—instead, it exists as a dynamic mixture where ammonia continuously shifts between its dissolved state and its ionized form. Because of this behavior, ammonium hydroxide is often represented symbolically rather than as a discrete molecule, making it more of a functional concept than a rigid chemical identity. Its usefulness lies in the strong basic environment it creates and the ease with which it releases ammonia vapors, giving it a characteristic, pungent smell commonly associated with household cleaners and industrial formulations.
Behavior of Ammonia in Water and Resulting Properties
Once dissolved, ammonia interacts with water in a way that produces a mildly alkaline solution. Only a fraction of the dissolved ammonia converts into ammonium and hydroxide, meaning the solution remains relatively weak compared to strong bases. This mildness, combined with its volatility, gives ammonium hydroxide several characteristic features: it releases a strong odor, it evaporates quickly from open surfaces, and it produces a cooling sensation when exposed to air as dissolved ammonia escapes. The balance between dissolved ammonia and its ionized form is influenced by temperature, concentration, and exposure to the atmosphere. This shifting nature is central to understanding why the solution behaves the way it does and why it cannot be concentrated indefinitely. In vector illustrations, this mixture is often shown as a container of liquid with ammonia gas above it, arrows indicating dissolution, and simplified ions drifting within the water to emphasize the fluid, reversible interaction that defines the solution.
Chemical Characteristics and Reactivity in Solution
Ammonium hydroxide acts as a base because it increases the presence of hydroxide ions in water, allowing it to neutralize acids and participate in reactions typical of alkaline solutions. It can precipitate certain metal ions from solution, form coordination complexes with transition metals, and alter the chemical environment in mixtures that depend on pH adjustments. Its ability to release ammonia vapor makes it highly effective in cleaning products, where the gas helps lift oils, break down residues, and dissolve certain types of stains. When heated or left open, the solution gradually loses ammonia, becoming weaker over time. This characteristic must be considered in laboratory or industrial contexts where strength and consistency are important. In vector diagrams, these behaviors are often communicated through stylized arrows indicating gas release, liquid mixing, or ion formation inside the solution.
Applications in Household, Industrial, and Laboratory Settings
Ammonium hydroxide is widely used in both everyday and technical environments. In household cleaners, it acts as a powerful degreasing agent, capable of cutting through oils, grime, and soap residues on surfaces such as glass, tiles, and metal fixtures. In industrial settings, it plays a role in textiles, rubber processing, water treatment, and chemical manufacturing, where its basic properties help regulate reactions or modify the characteristics of raw materials. Laboratories rely on ammonium hydroxide for pH control, metal precipitation tests, and preparation of buffer solutions. Its role in complex ion formation also makes it useful in qualitative analysis for identifying metal ions based on color changes and precipitate formation. This combination of versatility, accessibility, and effectiveness is why ammonium hydroxide remains a staple across so many different fields.
Physical Appearance and Sensory Characteristics
In its typical form, ammonium hydroxide appears as a clear, colorless liquid with a sharp, penetrating smell due to the continual presence of ammonia gas. The odor is strong enough to be detected even at low concentrations and can become irritating at higher levels, which is why proper ventilation is important when the solution is used indoors. The liquid feels slippery to the touch, reflecting its alkaline nature, and it can irritate skin or eyes upon contact. In its strongest commercial versions, the solution releases vapors rapidly, forming visible fumes in humid environments. Vector illustrations often represent this by showing gaseous movement above a liquid surface, as well as simple icons indicating ventilation or caution, helping communicate both its usefulness and its need for careful handling.
Representation in Vector Illustration and Conceptual Diagrams
Vector illustrations of ammonium hydroxide typically highlight the dynamic relationship between dissolved ammonia and the liquid water environment. A common approach is to depict the solution inside a beaker or container, with evaporating ammonia rising from the surface and simplified ions dispersed in the liquid to represent the interaction that gives the solution its alkaline behavior. Some illustrations also depict reaction scenarios, such as how metal ions respond to the solution, or more abstract chemical visuals that show ammonium and hydroxide ions arranged conceptually. These diagrams help clarify that ammonium hydroxide is not a fixed molecular entity but a fluid, shifting system defined by equilibrium rather than a single structural formula.