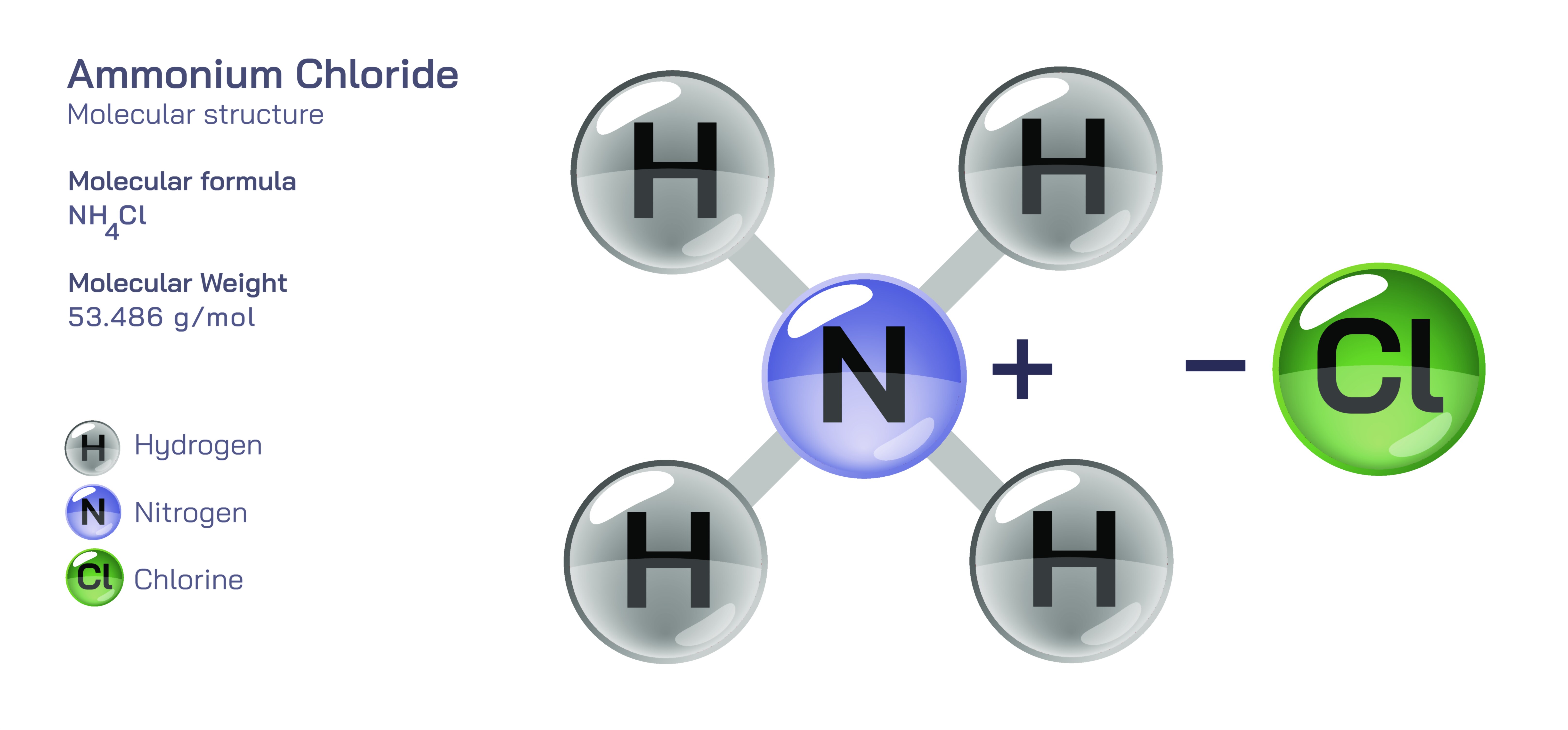
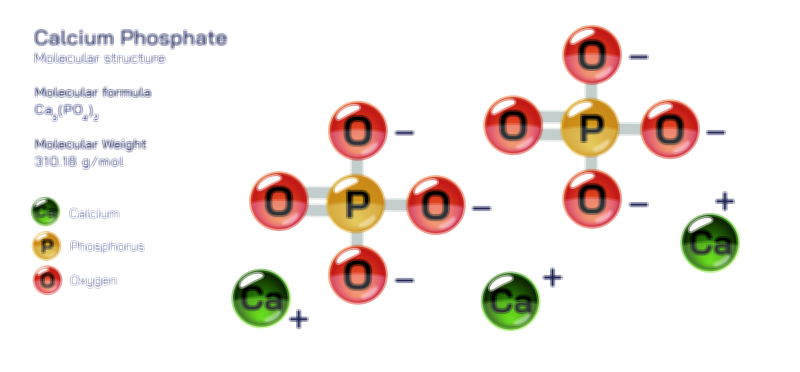
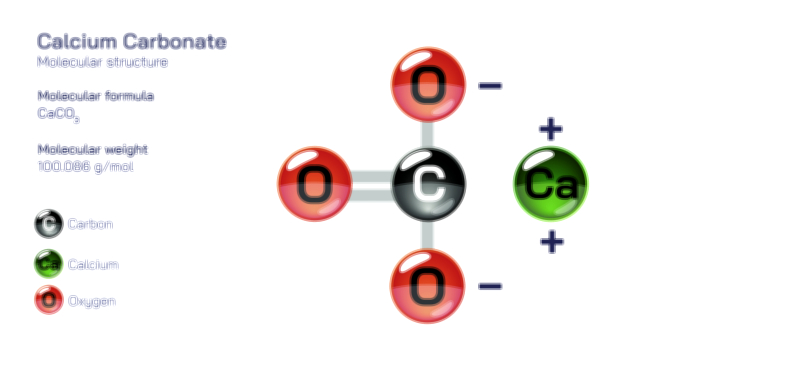
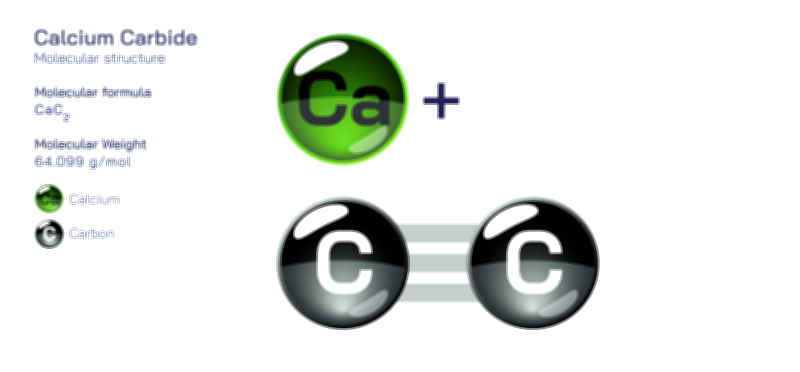
Ammonium Chloride — Structure, Properties, and Chemical Behavior
Understanding the Nature of Ammonium Chloride
Ammonium chloride is a widely known inorganic salt valued for its stability, versatility, and usefulness across both industrial and biological applications. It appears as a white, crystalline material with a sharp, cooling taste and dissolves readily in water, producing a mildly acidic solution. The compound forms when ammonia and hydrogen chloride interact, creating a material that balances simplicity with remarkable functional range. Even though its structure belongs to the category of ionic solids, ammonium chloride behaves in ways that set it apart from many other salts. It sublimes rather than melts under strong heat, transforming directly from solid to vapor and releasing the components that formed it. This behavior reflects a subtle internal arrangement held together by ionic forces that are strong enough for stability yet responsive enough to break apart under heat. A conceptual or vector-style illustration of ammonium chloride typically presents a neatly arranged lattice of positively charged ammonium groups and negatively charged chloride ions, symbolizing the order and balance that define its identity.
The Ammonium Component and Its Influence on the Compound
At the heart of ammonium chloride lies the ammonium group, which brings a unique character to the salt. Ammonium is a positively charged cluster formed when ammonia binds an extra hydrogen, creating a stable, symmetric structure. This group behaves similarly to metal cations in salts, yet its origin in a common gas gives it a distinctive personality. In ammonium chloride, the ammonium group contributes acidity, solubility, and reactivity to the compound. It interacts strongly with water, forming an acidic environment that influences how the salt behaves in biological and chemical systems. This acidity explains why the compound is commonly used in certain medical preparations and in laboratory reactions requiring a controlled acidic influence. In visual representations, ammonium is often shown as a compact central unit surrounded by hydrogen markers, emphasizing its balanced shape and ability to pair with chloride ions across a structured lattice.
The Chloride Ion and Its Functional Role
Chloride is one of the most familiar and stable ions in chemistry, contributing reliability and solubility to the compounds it forms. In ammonium chloride, the chloride ion acts as the stabilizing partner to ammonium, creating the ionic attraction that holds the solid together. Its presence ensures that the compound dissolves easily in water, generating a clear solution used in countless laboratory and industrial contexts. Chloride’s chemical calmness paired with ammonium’s reactivity gives the salt a blend of characteristics: steady enough to store safely, yet responsive enough to participate in many useful reactions. Over time and under normal conditions, the chloride ion does not significantly react or degrade within the salt, contributing to ammonium chloride’s long shelf life and trusted behavior.
Physical Characteristics and Thermal Behavior
Ammonium chloride forms fine crystals or granules that remain free-flowing under dry conditions. Its solubility in water increases with heat, making it easy to prepare concentrated solutions for scientific or industrial use. One of its most interesting physical traits is its ability to sublime when strongly heated, bypassing the liquid state entirely. During sublimation, the compound breaks apart into ammonia and hydrogen chloride vapors, which recombine into the solid when cooled. This reversible transformation demonstrates the delicate balance inside the crystal structure and is often used in teaching environments to visually illustrate the behavior of ionic solids under thermal stress. The compound remains stable at room temperature but must be kept dry because moisture can cause clumping or create undesired reaction conditions during storage.
Chemical Behavior and Reactivity in Different Environments
In water, ammonium chloride produces an acidic solution because the ammonium component releases hydrogen into the surrounding environment. This mild acidity affects how metals, biological tissues, and other chemicals respond when exposed to the solution. The compound also serves as a valuable source of nitrogen in reactions and industrial formulations. Its predictable dissociation in water makes it a reliable reagent in buffer preparations, where it helps maintain controlled acidity levels. When mixed with strong bases, the solution releases ammonia, a behavior used in laboratories for identifying ions or adjusting chemical conditions. The chloride component acts as a counterbalance during such reactions, remaining largely unchanged while ammonium participates in chemical shifts. These combined behaviors give ammonium chloride a versatile role across scientific and industrial domains.
Role in Medicine, Industry, and Food Processing
Ammonium chloride’s range of applications demonstrates how its structure and behavior translate into practical usefulness. In medicine, it has been used in treatments requiring increased acidity in the body or as an expectorant to help loosen mucus. In industrial settings, the compound appears in metalworking, where it cleans surfaces and assists in preparing metals for bonding or coating. It is also used in dry cell batteries, particularly in older or specialty battery types where it helps conduct electrical charge within the cell. In food processing, ammonium chloride has a niche role as a flavoring agent, especially in traditional recipes from northern Europe where its salty, slightly bitter taste is valued. The compound also appears in dyeing, textile processing, and certain fermentation processes, demonstrating its usefulness across both organic and inorganic chemical systems.
Vector Illustration Representation of Ammonium Chloride
A vector illustration of ammonium chloride often emphasizes the balanced ionic structure that defines the compound. The ammonium portion is typically portrayed as a symmetrical cluster, while the chloride ion appears as a single, rounded unit paired nearby. Illustrations may also include lattice-like arrangements or geometric patterns to represent the crystalline structure, helping viewers understand how the ions align in solid form. Some educational diagrams highlight its dissolution in water by showing ions separating and dispersing evenly throughout the liquid. Such visuals help communicate the compound’s dual nature as a structured solid and a highly soluble ionic material, making its behavior and uses easier to grasp.