Ammonium Nitrate — Crystalline Oxidizer and Chemical Behavior
Understanding the Nature of Ammonium Nitrate
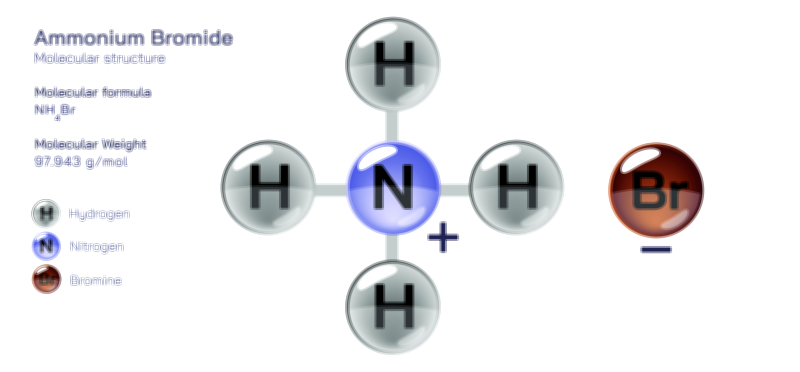
Ammonium nitrate is a widely known chemical compound valued for its strong oxidizing properties and its unique crystalline form that dissolves easily in water. It is composed of ammonium and nitrate groups that join together to create a solid material that is both highly useful and highly sensitive, depending on the conditions in which it is handled. In its pure form, ammonium nitrate appears as white granules or prills, which flow easily and store well in controlled environments. A vector illustration of ammonium nitrate usually shows its balanced pairing between the nitrogen–hydrogen cluster of the ammonium group and the oxygen-rich nitrate group, pairing them in a way that highlights how one side of the compound brings hydrogen and nitrogen while the other brings an abundance of oxygen. This oxygen-rich identity is the reason ammonium nitrate is considered a powerful oxidizer, capable of releasing large amounts of energy when combined with a suitable fuel or exposed to specific triggering conditions. Its structure is simple enough to represent clearly in diagrams, yet its behavior is complex enough to make it a carefully managed material across many industries.
Crystalline Structure and Physical Properties
The crystalline structure of ammonium nitrate is one of the features that make it so versatile and widely used. It naturally forms stable, odorless crystals that are highly soluble in water and tend to absorb moisture from the air. The compound can shift between several solid forms depending on temperature, giving it a distinct physical sensitivity that requires careful storage to avoid clumping or instability. These changes in crystal structure influence how easily the material flows and how it behaves in mixtures. In vector illustrations, the crystalline appearance is often represented through geometric patterns or repeated shapes that convey the orderly arrangement of particles. The nitrate portion, containing multiple oxygen atoms, brings oxygen-rich character to the compound, while the ammonium portion offers hydrogen that can interact with other materials under the right conditions. Together, these pieces create a crystal that is physically simple yet chemically energetic, a combination that explains both its usefulness and its sensitivity.
Role as a Powerful Oxidizing Agent
Ammonium nitrate’s most defining characteristic is its strength as an oxidizer. An oxidizer is a substance that supplies oxygen to fuel a reaction, and ammonium nitrate stores oxygen in a form that can be released rapidly when triggered. On its own, the compound is stable under controlled conditions, but when mixed with certain fuels or subjected to heat and confinement, the oxygen it contains becomes available for rapid reaction. This makes ammonium nitrate a foundation for many industrial and mining explosives, though only when purposefully combined with fuel sources to create stable, regulated products. In less controlled scenarios, improper handling, contamination, or overheating can cause the material to release energy quickly, sometimes with hazardous results. This dual nature—useful when controlled, dangerous when mishandled—makes ammonium nitrate a compound that must always be treated with knowledge and respect. In vector illustrations, this oxidizing ability is often portrayed through directional arrows or oxygen-emphasizing graphics that show how its structure supplies oxygen during reactions.
Agricultural Importance and Fertilizer Applications
Despite its well-known energetic potential, ammonium nitrate remains one of the most effective nitrogen fertilizers used worldwide. It provides plants with two forms of nitrogen at once: one part that acts quickly and another part that acts gradually, making it extremely beneficial for crops that require sustained nutrient availability. Because nitrogen is a key component of plant growth, contributing to leaf development and overall vitality, ammonium nitrate plays a central role in agricultural productivity, particularly in regions that depend on consistent nutrient supply. Its water solubility ensures that nutrients reach plant roots efficiently, while its predictable behavior in soil makes it relatively easy to manage when applied correctly. In agricultural vector illustrations, the compound is often shown as small granules dissolving into soil, with arrows representing nutrient movement toward the roots, making it easy to visualize how the fertilizer distributes its nitrogen over time.
Thermal Sensitivity and Decomposition Behavior
Ammonium nitrate is stable under ordinary conditions, yet it responds strongly to heat. When warmed gradually in an open environment, it breaks down into harmless gases. However, if subjected to high heat or confined pressure, the decomposition becomes much more energetic, releasing a large volume of gas in a very short time. This fast expansion is what gives ammonium nitrate its explosive hazard potential when mishandled. It does not burn by itself, but in certain conditions it can react violently by supplying the oxygen needed for combustion. Contaminants such as oils, metals, or organic materials can further destabilize it by creating uneven reaction zones. This sensitivity is the reason strict global regulations exist regarding its storage, transport, and use. In vector illustrations, thermal sensitivity is often portrayed using heat waves or expanding shapes, helping convey how temperature changes influence the compound’s stability.
Industrial, Mining, and Safety Contexts
Industrially, ammonium nitrate plays a crucial role in controlled explosive formulations used in mining, construction, and quarrying. In this context, it is combined with fuel sources in highly regulated mixtures designed for predictable performance. These mixtures are engineered to be safe during handling but effective when properly activated. Outside industrial use, however, ammonium nitrate must be handled with vigilance to prevent accidental heating, contamination, or confinement. Safety guidelines emphasize ventilation, separation from combustible materials, and avoidance of moisture, which can cause clumping and uneven heating. The compound’s history includes both beneficial and tragic moments, showing how powerful chemistry can be when controlled—and how dangerous it becomes when misused. Visual diagrams in this context often illustrate the difference between safe granular storage and the hazardous conditions that lead to rapid decomposition.
Representing Ammonium Nitrate Through Vector Illustration
A vector illustration of ammonium nitrate typically captures its dual identity. It may show the ammonium portion and the oxygen-rich nitrate portion in simplified symbolic forms to highlight their connection. Illustrations may also depict crystalline granules, flowing prills, nutrient release in soil, or oxygen-supporting behavior in reactions. These visual elements help communicate how a single compound can serve agricultural needs, industrial purposes, and scientific studies while requiring careful management to remain safe. The strength of a good vector illustration lies in its ability to take a chemically energetic material and represent it in a clear, approachable, and informative way.