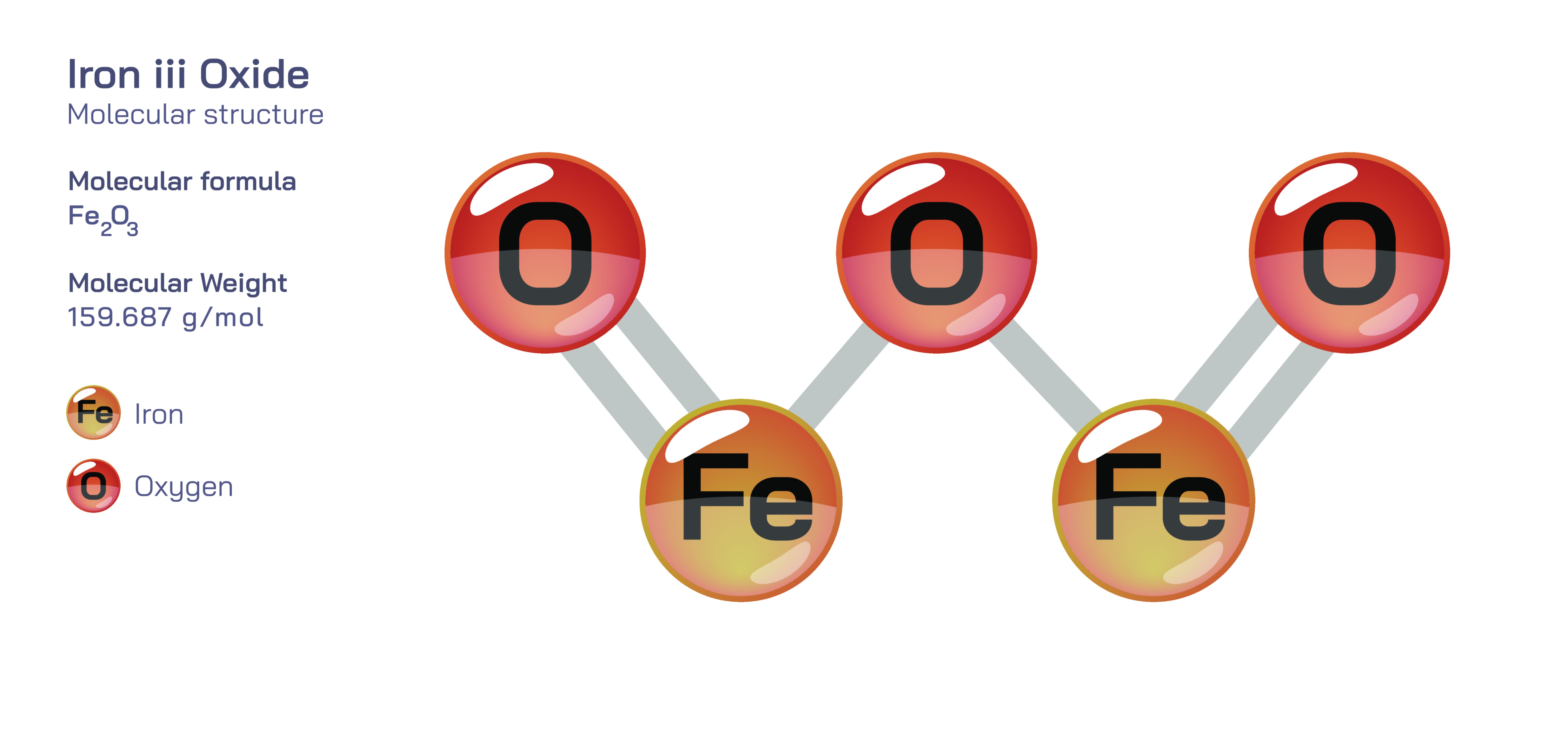
Iron(III) Oxide — Rust Formation, Chemical Structure, and Corrosion Process in Metals
Iron(III) oxide is one of the most familiar chemical compounds in daily life, not because people encounter it in scientific laboratories but because it appears as rust, the reddish-brown flaky layer that forms on iron and steel when exposed to air and moisture. Although rust may seem like a simple stain on metal, it actually represents a complex chemical transformation that changes iron from its metallic elemental state to a stable oxide. Understanding this transformation is essential in chemistry education because it shows how environmental factors can alter the atomic structure of elements and produce entirely new substances with very different properties. In its pure form, Iron(III) oxide is a crystalline solid commonly known as hematite, and it occurs naturally in rocks and soils, but when students encounter it through the rusting of everyday objects, it becomes an accessible and relatable gateway to learning about oxidation, corrosion, and chemical reactions.
In rust formation, iron loses its identity as a shiny, silvery metal and gradually transforms into a brittle compound that no longer carries the desirable strength and durability that make iron and steel useful in construction, machinery, tools, and transportation. This transformation begins when iron comes into contact with oxygen in the air, but the process only accelerates significantly when moisture is present. Water allows oxygen to interact with iron more efficiently by enabling the movement of charged particles on the metal surface. As the process continues, metal gradually converts into Iron(III) oxide, which separates from the underlying metal as a crumbly surface layer. This layer does not protect the inner metal the way the oxide layers of aluminum or chromium do; instead, it flakes away, exposing a fresh surface underneath that continues to react. As a result, rust spreads progressively deeper until the metallic structure is weakened, eventually leading to structural failure.
The atomic nature of Iron(III) oxide explains its behavior during corrosion. When iron reacts with oxygen and moisture, its atomic configuration shifts from a solid lattice of metallic atoms to a new arrangement consisting of iron atoms bonded with oxygen atoms. This change rearranges the electrons in the outer shells, giving the new compound very different physical characteristics compared with the original metal. Iron becomes brittle rather than strong, porous rather than solid, and reddish-brown rather than metallic gray. In educational diagrams, this idea is often shown using molecular models that depict iron atoms surrounded by oxygen atoms in a repeating lattice. The representation helps students visualize that rust is not simply damaged metal but an entirely different compound that no longer has the mechanical strength required in engineering applications.
Rusting is not only a scientific observation but an economic challenge. Bridges, ships, rails, automobiles, household tools, and underground pipelines require ongoing protection against corrosion. Prevention is therefore almost as educationally important as the rusting process itself. Although prevention is not a required part of the classroom definition of Iron(III) oxide, understanding how rust develops makes it easier to appreciate why metals are painted, coated with oil, galvanized with zinc, or alloyed with elements such as chromium to form stainless steel. These approaches slow or stop the access of oxygen and moisture, protecting the metal underneath and restricting the formation of Iron(III) oxide. Students observing real objects in their environment—such as an unmaintained gate rusting while a coated gate remains intact—can visually understand how chemistry applies to real-life technological challenges.
The rusting process is also deeply connected to environmental science. Objects rust more quickly in humid regions than in dry climates because moisture speeds oxidation. Saltwater accelerates rust even further, which explains why ships and coastal structures corrode at a much faster rate than those in inland areas. Corrosion therefore becomes not only a chemistry lesson but a geographical and engineering one, helping learners recognize why specific materials are chosen for specific environments. When educational graphics show Iron(III) oxide forming faster in the presence of saltwater droplets than plain rainwater, they reinforce this connection between chemical behavior and environmental conditions. It illustrates that the world is full of interacting variables, and even simple compounds follow patterns governed by chemistry.
Another important connection is how rusting reveals the flow of scientific logic. Iron(III) oxide forms because it is more stable than metallic iron under atmospheric conditions. The universe favors stability, and chemical reactions move atoms toward arrangements that require less energy over time. Rusting, therefore, becomes a natural pathway for iron to reach a more stable form, even if it comes at the cost of durability and strength from a human standpoint. In an educational setting, this reinforces a powerful scientific theme: materials do not remain static, and their transformation is guided by predictable rules of matter rather than random change. Rust, in this sense, becomes a demonstration of both the fragility of engineered materials and the enduring consistency of chemical processes.
Iron(III) oxide as a compound stretches far beyond rusted fences or neglected tools. In its purified mineral form, it has numerous industrial applications, including use as a pigment, as a polishing agent for lenses and glass, and as a component in magnetic storage materials. Even students who primarily learn about Iron(III) oxide through corrosion benefit from understanding that the same compound appears across different industries, demonstrating that materials behave differently and find different uses depending on context, purity, and form. A chemistry education illustration that shows both rusting metal and the crystalline form of hematite teaches that the identity of a compound remains uniform even when the circumstances of its formation make it seem undesirable in one case and useful in another.
At its deepest educational level, Iron(III) oxide highlights the connection between the atomic world and everyday life. Every time a bicycle chain rusts, every time a metal railing discolors in the rain, and every time steel loses its strength through neglect, students are watching a chemical transformation unfold in real time. Technology, chemistry, physics, materials science, and environmental studies meet at the surface of iron as it reacts with oxygen and moisture. When students understand the atomic structure of Iron(III) oxide, the rusting of metal stops being an accident or an inconvenience and becomes a predictable scientific reaction governed by the identity of atoms and the conditions of the environment.
Iron(III) oxide therefore serves as one of the most relatable and valuable learning tools in chemistry education. It anchors understanding not just in textbooks but in the world around us, showing that chemical change is constant, that materials evolve under environmental influence, and that scientific knowledge is essential for designing safer structures, more durable materials, and better solutions for protecting the objects we rely on. Where rust might appear as simple damage to the untrained eye, to a scientifically informed mind it becomes evidence of the laws of nature operating both invisibly and predictably.