Boyle’s Law — Temperature and Pressure Connection
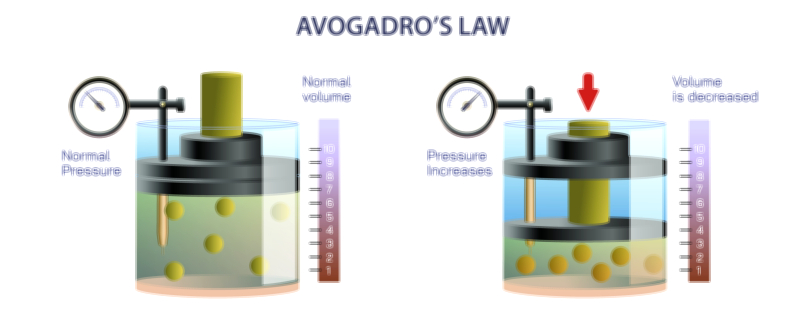
Boyle’s Law describes one of the most essential behaviors of gases, focusing on how their pressure changes when their volume shifts under conditions where temperature remains stable. Even without using mathematical expressions, the idea can be understood as the natural tendency of gas particles to spread out when given more space and become more forceful when confined to smaller spaces. Gas particles move constantly, colliding with the walls of whatever container they occupy, and these collisions create what we understand as pressure. When the available space becomes smaller, the same number of particles must move within a tighter region, increasing how often they strike the container’s walls and therefore increasing the pressure. When the space increases, those collisions become less frequent, leading to a decrease in pressure. What makes Boyle’s Law elegant is how predictable and consistent this relationship is when the temperature is held steady, allowing scientists and engineers to understand and control gas behavior with confidence.
The relationship between pressure and volume becomes particularly significant when considering temperature’s role in maintaining the stability of this pattern. Boyle’s Law applies only when the temperature is kept constant, meaning the internal motion of the gas particles does not speed up or slow down due to heating or cooling. Temperature directly affects particle motion, so if it were allowed to change, the pressure would also change for reasons unrelated to volume. By holding temperature fixed, the law isolates a pure interaction between pressure and volume, enabling a clear understanding of how gases respond when compressed or expanded. This connection emphasizes the importance of stable thermal conditions in scientific experiments and industrial processes, ensuring that measurements reflect true changes in spatial constraints rather than unintended temperature effects. In this way, Boyle’s Law becomes a foundational tool for analyzing gas behaviors in controlled settings.
The practical usefulness of Boyle’s Law appears in countless everyday and technological situations. When a syringe is used, pulling the plunger back increases the internal volume and lowers the pressure, allowing external pressure to push fluid or air into the chamber. Scuba diving offers another vivid example, where the pressure that increases underwater forces the air in tanks and lungs to occupy smaller volumes, making divers acutely aware of how pressure changes influence breathing and equipment. In automotive engineering, the compression of air-fuel mixtures in engines relies on the predictable behavior described by Boyle’s Law, ensuring efficient ignition and smooth performance. Even simple objects like balloons demonstrate this concept as the air inside adjusts to squeezing, stretching, or temperature shifts around it. Each example reflects how deeply this law is embedded not just in theory but in normal human experience.
Understanding Boyle’s Law also provides valuable insight into how gases function in natural environments. Atmospheric pressure decreases at higher altitudes because the air has more space to expand as the weight of the atmosphere above becomes lighter. This change in pressure influences breathing, weather patterns, and the behavior of clouds as air masses shift, expand, or compress through atmospheric movements. Meteorology, aviation, and environmental science rely on these predictable pressure-volume relationships to forecast weather, design safe aircraft cabins, and understand how air behaves under different conditions. The pressure variations inside the lungs during inhalation and exhalation also highlight how living organisms make use of these same principles, with expansion and compression driving airflow that sustains life.
When thinking about the deeper meaning behind Boyle’s Law, it becomes clear that the law is not just a rule about gases but a reflection of the way matter behaves under constraints. Gases are the most responsive states of matter because their particles are free to move independently, making them excellent examples of how space and confinement influence physical behavior. The law serves as a reminder that the natural world follows ordered patterns, and these patterns can be observed, measured, and applied in ways that benefit science, technology, and daily living. It demonstrates how understanding the movement of invisible particles leads to real-world advancements, from medical devices to industrial machinery to environmental predictions.
Boyle’s Law remains a cornerstone of classical gas theory because of its clarity and its ability to bridge intuition with scientific depth. Anyone who has squeezed a rubber ball, pumped a bicycle tire, or felt their ears adjust during a flight has experienced the effects described by the law. It takes these familiar sensations and transforms them into a formal explanation of how pressure and volume interact under stable temperature conditions. Without relying on equations, the essence remains straightforward: when the space available to a gas changes, its pressure changes in a predictable and opposite direction, provided the temperature stays the same. This simple yet powerful idea continues to shape scientific understanding, engineering design, and practical decision-making across many fields.