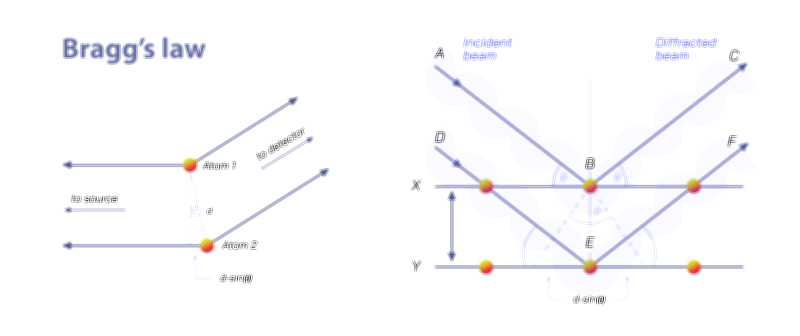
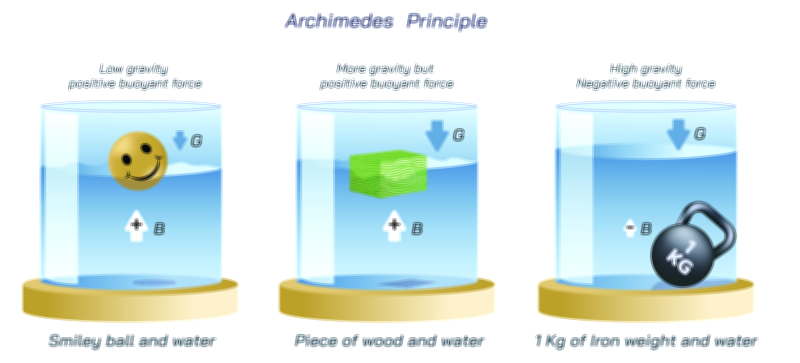
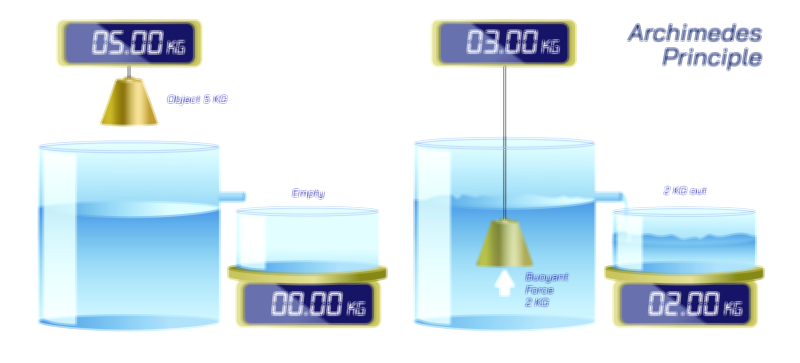
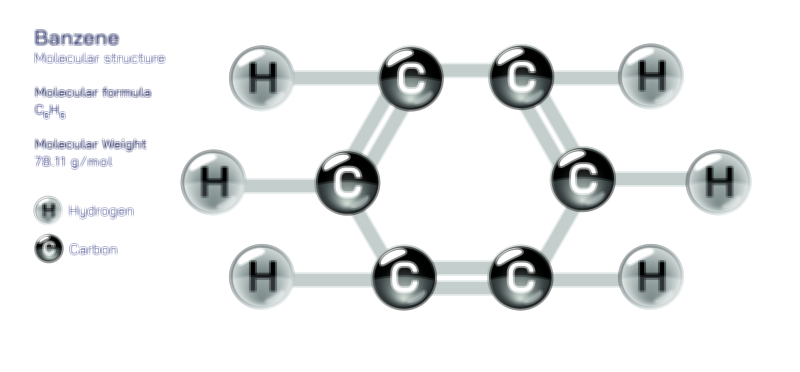
Avogadro’s Law — Understanding the Gas Volume Relationship in Fundamental Chemistry
The Central Idea Behind Avogadro’s Law
Avogadro’s Law describes one of the most important relationships in the study of gases, offering a simple yet powerful explanation of how gas volume changes when the amount of gas varies. At its core, the law states that equal volumes of gases contain equal numbers of particles when they are at the same temperature and pressure. This idea highlights how gases behave in a remarkably consistent way, regardless of their chemical identity. Whether the particles belong to a light gas or a heavier one, their collective behavior reveals a shared tendency to spread out and occupy space in proportion to the number of particles present. Avogadro’s insight provides a foundation for understanding why gases expand, compress, and interact so predictably, forming a key pillar in the study of physical chemistry and thermodynamics.
How Gas Particles and Volume Are Connected
The essence of Avogadro’s Law lies in how gas particles naturally move. Unlike solids and liquids, where particles remain closely packed, gas particles are free to move in all directions, constantly spreading apart from one another. When more particles are added to a container while temperature and pressure remain unchanged, these particles occupy the available space and increase the volume the gas requires to maintain stable conditions. This direct connection between the number of gas particles and the space they fill is what Avogadro recognized. Every gas, under the same conditions, behaves as though each particle needs a consistent amount of room to move. This explains why gas samples with different chemical identities can still follow the same relationship between particle count and volume.
Why Avogadro’s Law Helps Us Understand Gas Behavior
Avogadro’s Law offers an elegant way to explain everyday observations about gases. It clarifies why inflating a balloon makes it expand, since additional gas molecules force the balloon to stretch outward. It also explains why heating a gas-filled container causes expansion—because the warmer particles move more vigorously and require more space. Although these effects involve temperature changes, the underlying principle remains the same: the amount of space occupied by a gas depends on the number of particles acting within it. This relationship is essential for interpreting gas-related phenomena such as diffusion, pressure changes, and volume adjustments during chemical reactions. Without this understanding, the predictable behavior of gases would appear chaotic rather than orderly.
How Avogadro’s Law Helps Compare Different Gases
Another powerful aspect of Avogadro’s Law is that it allows all gases to be compared on equal terms. Even though gases differ in their masses, densities, and chemical properties, they all follow the same volume relationship when temperature and pressure are matched. This means that a container filled with helium and another filled with oxygen at identical conditions will hold the same number of particles, even though the gases differ dramatically in weight and behavior. This insight allows scientists to study gases based on their particle count rather than relying solely on mass, making it possible to standardize measurements and perform consistent comparisons across countless substances. The shared behavior of gases under the same conditions reflects a deeper unity in the way particles respond to energy and space.
The Law’s Role in Understanding Chemical Reactions and Gas Mixtures
Avogadro’s Law is especially important in understanding chemical reactions that involve gases. Because gases expand to fill available space in a predictable way, scientists can use their volumes to determine how much of one gas will combine with another during a reaction. When gases react or are produced, the resulting volumes reflect the proportions of particles involved. This makes it possible to analyze reactions without needing to observe the particles themselves. The law also helps explain how gas mixtures behave. When multiple gases are placed in the same container, each spreads out independently, and their combined volume reflects the total number of particles, not their chemical differences. These ideas help build the framework for understanding air composition, respiratory processes, industrial gas applications, and atmospheric science.
Why Avogadro’s Law Supported the Concept of the Mole
The development of the mole—a unit used to describe the amount of a substance—was strongly supported by Avogadro’s ideas. Because gases behave consistently at the particle level, Avogadro’s work allowed scientists to estimate the number of particles in a given volume under standard conditions. This paved the way for defining a consistent unit that represents a specific number of particles. While Avogadro himself did not establish the modern definition, his insights made it possible for later scientists to connect particle quantity, mass, and gas volume into a unified measurement concept. The mole is now one of the most fundamental units in chemistry, used in every level of study, from laboratory reactions to biological processes.
The Broader Importance of Avogadro’s Law in Science
Avogadro’s Law forms part of a broader collection of gas principles that help explain the behavior of matter in its most flexible and dynamic state. By highlighting the connection between particle quantity and space, the law gives scientists a way to predict, analyze, and manipulate gases with remarkable accuracy. Whether used in medicine, environmental monitoring, engineering, or chemical manufacturing, this understanding ensures that gas-related processes remain stable, efficient, and predictable. It also provides a window into the fundamental nature of matter, revealing how even invisible particles operate under universal physical patterns that shape everything from weather systems to combustion engines. Appreciating Avogadro’s Law deepens our understanding of the harmony between microscopic particles and macroscopic behavior, showing how simple principles govern complex natural systems.