Beer Lambert Law — Absorbance and Concentration Relationship
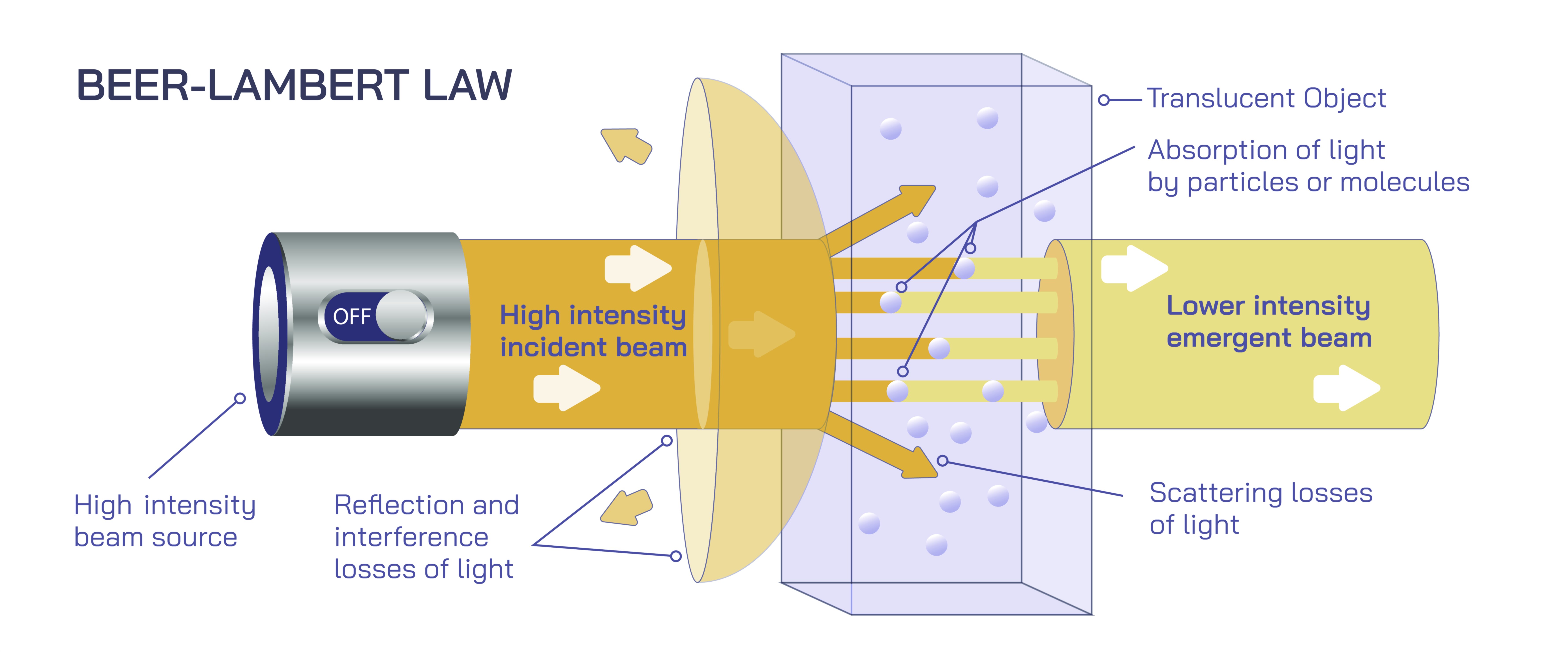
The Beer Lambert Law describes one of the most fundamental relationships in analytical chemistry, explaining how the intensity of light passing through a substance changes in relation to the characteristics of that substance. Even without using mathematical expressions or symbolic formulas, this law can be understood as a guiding principle that connects how much light is absorbed by a sample to how concentrated that sample is. When a beam of light enters a solution or material, some of that light is taken up by the particles present within it. The more particles there are, the more opportunities they have to interact with the incoming light, causing a greater reduction in its intensity. This direct connection makes the Beer Lambert Law invaluable for scientists and industries that need to determine concentrations without physically altering or consuming the samples they analyze. It transforms light itself into a precise measurement tool, allowing users to understand the makeup of substances through controlled illumination.
At its core, the law rests on the straightforward idea that absorbance rises when concentration rises, because a more crowded environment of molecules or ions blocks or diminishes more of the incoming light. Absorbance also increases when the path the light travels through is longer, because the light encounters more matter along its journey. Though no numerical relationships are expressed here, the concept remains clear: thicker samples or more concentrated samples absorb more light, while thinner or more diluted ones absorb less. This makes the Beer Lambert Law a foundational principle in laboratories because it provides a predictable pattern that matches what happens in real-world measurements. Instruments designed to analyze absorbance rely heavily on this concept, and their construction reflects the need for stability, alignment, and consistency in the way light interacts with the sample. When absorbance values are measured, the results reveal the internal properties of the sample in a non-destructive and repeatable way.
Understanding the meaning of absorbance helps illustrate why this law is so important. Absorbance is essentially a measure of how much energy is taken in by the sample rather than passing through it. Different substances absorb light differently depending on their structure, the wavelengths involved, and the physical conditions of the measurement. In a biochemical or environmental setting, molecules may possess unique patterns of light absorption, much like an optical fingerprint that identifies them. By using a single beam or a dual-beam light source, instruments known as spectrophotometers compare the incoming and outgoing intensities to determine absorbance. Because the Beer Lambert Law assures that absorbance rises consistently with concentration under proper conditions, researchers can build reliable calibration curves and compare unknown samples against them. This transforms absorbance data into meaningful concentration values, turning light measurements into chemical insights.
The law is widely applied in fields such as environmental monitoring, pharmaceuticals, clinical diagnostics, food science, and chemical manufacturing. For example, it allows laboratories to monitor pollutant levels in water by measuring how much light is absorbed by dissolved contaminants. In medical diagnostics, it helps determine concentrations of biomolecules in blood or urine without requiring invasive procedures or complicated separation steps. In the food and beverage industry, it enables quality control by verifying ingredient strength, color intensity, and purity. Even in research settings, the Beer Lambert Law aids scientists in tracking reaction progress, studying molecular behavior, and evaluating how substances interact under changing conditions. Its reliability has made it one of the most widely used principles in both academic and industrial laboratories because it offers a combination of speed, accuracy, and non-destructive testing.
However, the Beer Lambert Law depends on specific conditions to function properly, and understanding these conditions deepens appreciation for the precision of the method. The light used must consist of a narrow, well-defined wavelength so that variations in absorption do not interfere with the relationship. The sample must be uniform, meaning the particles or molecules must be evenly distributed so that light interacts with them in a consistent manner. The container holding the sample must have clear, parallel surfaces to prevent reflection or scattering that could distort the absorbance reading. When samples become too concentrated, the particles may interact with each other, or they may scatter light rather than absorb it, weakening the reliability of the relationship. Similarly, colored impurities or bubbles in the solution can interfere with the accuracy, which is why careful preparation and consistent handling are essential for measurements based on this law.
Beyond practical usage, the conceptual significance of the Beer Lambert Law lies in the elegant way it links physical interactions with measurable outcomes. Light, a form of energy that moves freely through transparent materials, becomes a sensitive probe when directed through a sample. The way that sample diminishes or alters the light carries information about its internal makeup. This transformation of an invisible process into readable data is the essence of analytical chemistry, turning natural phenomena into useful technological tools. The law symbolizes the harmony between theoretical understanding and practical measurement, making it not only a tool for laboratory work but also a model for scientific thinking. It shows how predictable patterns in nature can be harnessed to create methods that improve health, industry, and environmental stewardship.
In everyday life, people encounter the effects of absorption without realizing it, such as when tinted glass reduces brightness or when colored liquids appear darker as they become more concentrated. The Beer Lambert Law formalizes this everyday experience into a precise scientific rule that allows professionals to work with confidence and precision. Even though this explanation avoids the mathematical details, the conceptual clarity remains intact: more substance leads to more absorption, and this absorption reveals valuable information. Through this simple but powerful connection, the Beer Lambert Law continues to be a cornerstone of scientific analysis, enabling progress, discovery, and control across countless disciplines.